Use data from Table 13.8 and Figure 13-10 to estimate the temperature at which K = 1.0
Question:
Use data from Table 13.8 and Figure 13-10 to estimate the temperature at which K = 1.0 x 106 for the reaction
![]()
Table 13.8
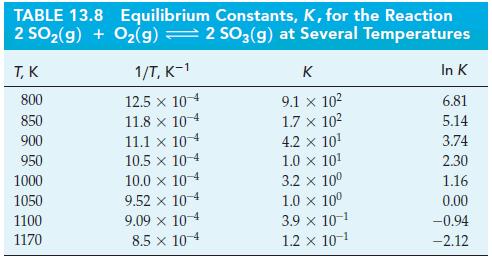
Figure 13-10
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:





