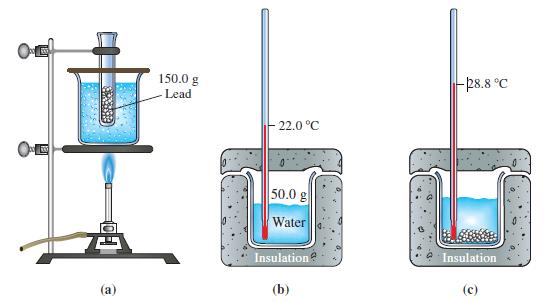
Use data presented in Figure 7-3 to calculate the specific heat capacity of lead. Figure 7-3 OE
Question:
Use data presented in Figure 7-3 to calculate the specific heat capacity of lead.
Figure 7-3
Transcribed Image Text:
OE 200 (a) 150.0 g -Lead 22.0 °C 50.0 g Water Insulation (b) -28.8 °C Insulation C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
Analyze Keep in mind that if we know any four of the five quantitiesq m specific heat cap...View the full answer

Answered By

PRINCE PANDEY
I am Indian Chartered Accounting having a strong hold in the subjects of Accounting, IFRS Reporting, Indian
Taxation, Cost Accounting, Auditing. I have vast experience of teaching a student with easy way problem-solving approach.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Refer to Example 7-2. The experiment is repeated with several different metals substituting for the lead. The masses of metal and water and the initial temperatures of the metal and water are the...
-
Prepare a graph of specific heat capacities for metals versus their atomic weights. Combine the data in Figure 5.4 and the values in the following table. What is the relationship between specific...
-
The specific heat capacity of silver is 0.24 JoC-1g-1. a. Calculate the energy required to raise the temperature of 150.0 g Ag from 273 K to 298 K. b. Calculate the energy required to raise the...
-
Part C A 100.0 ? and a 150.0 ? resistor, both ratedat 2.00 W, are connected in series across a variable potentialdifference. What is the greatest this potential difference can bewithout overheating...
-
Refer to the information in Problem 3-5B. Assume that Foster uses the FIFO method to account for its process costing system. The following additional information is available. Beginning goods in...
-
When during a jump is your speed zero?
-
In 1951, DuPont began using the chemical perfluorooctanoic acid to manufacture Teflon. Due to the dangerous nature of the chemical, DuPont was given special instructions by its supplier to dispose of...
-
On July 31, 2014, Redeker Company had a cash balance per books of $6,140. The statement from Nashota State Bank on that date showed a balance of $7,690.80. A comparison of the bank statement with the...
-
Tina is aware that thousands of children starve to death every day, but only after reading a story about a single starving child did she decide to donate money to a charitable organization. Tina's...
-
The G. Saussy Manufacturing company is putting out four new electronic components. Each of Saussy's four plants has the capacity to add one more product to its current line of electronic parts. The...
-
How much heat is required to raise the temperature of 100.0 mL of water (approximately 100.0 g) from room temperature, typically 21.0 C, to body temperature, typically 37.0 C? (Assume the specific...
-
In the neutralization of a strong acid with a strong base, the essential reaction is the combination of H + (aq) and OH - (aq) to form water. Two solutions, 25.00 mL of 2.50 M HCl(aq) and 25.00 mL of...
-
Why did Amazon decide to get into cloud computing? This business is radically different from shipping books and other physical products. Do you think Amazon should continue to keep AWS as part of...
-
The following table shows historical end-of-year prices for a stock. What is the Arithmetic average return? What is the Standard deviation? A B 1 Year Stock price 2 1 38.44 3 2 43.05 4 3 30.14 5 4...
-
Suppose you have 1000 in your bank account. The price of wheat is currently 100 per barrel. Due to unfavourable weather conditions you believe that supply will fall in the near future, however...
-
A bond with a $1,000 par, 4 years to maturity, a coupon rate of 4%, and annual payments has a yield to maturity of 4.1%. What will be the actual percentage change in the bond price if the yield...
-
1. In its most recent financial statements, Del-Castillo Inc. reported $70 million of net income and $850 million of retained earnings. The previous retained earnings were $830 million. How much in...
-
What is the yield to maturity of a 5-year, 7.5% coupon rate $1000 par value bond priced currently at $1,010? % Place your answer in percentage form using two decimal places. Do not use the percent...
-
Hamlin Company has purchased land and a warehouse for $18,000,000. The warehouse is expected to last 20 years and to have a residual value equal to 10 percent of its cost. The chief financial officer...
-
Three forces with magnitudes of 70pounds, 40 pounds, and 60 pounds act on an object at angles of 30, 45, and 135, respectively, with the positive x-axis. Find the direction and magnitude of the...
-
Financial Statement Effect of Equity Securities Presented below are three unrelated situations involving equity securities. Situation 1 An equity security, whose market value is currently less than...
-
Equity Securities the Financial Accounting Standards Board issued accounting guidance to clarify accounting methods and procedures with respect to certain debt and all equity securities. An important...
-
Investment Accounted for under the Equity Method on July 1, 2011, Fontaine Company purchased for cash 40% of the outstanding capital stock of Knoblett Company. Both Fontaine Company and Knoblett...
-
A box of mass 2 . 0 kg is at rest on a tar ramp that is 3 7 degrees to the horizontal. 8 . 1 If the box slides down at constant velocity, what is the coefficient of friction? 8 . 2 If the box...
-
Microwave ovens rotate at a rate of about 5.2 rev/min. A)What is this in revolutions per second? B)what is the angular velocity of this microwave in radians per second
-
A frictionless pendulum clock on the surface of the earth has a period of 1.00 s. On a distant planet, the length of the pendulum must be shortened slightly to have a period of 1.00 s. What is true...

Study smarter with the SolutionInn App


