Use Hesss law to determine r H for the reaction 1 CO(g) + O2(g) CO(g),
Question:
Use Hess’s law to determine ΔrH° for the reaction
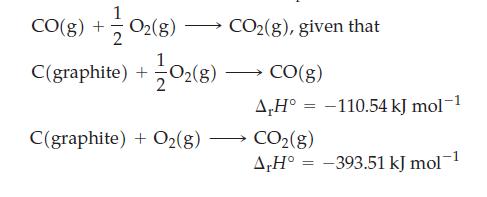
Transcribed Image Text:
1 CO(g) + O2(g) →→→→ CO₂(g), given that C(graphite) + +10₂(8) C(graphite) + O₂(g) CO(g) A,H° -110.54 kJ mol-1 CO₂(g) A,H° = -393.51 kJ mol-1 =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To use Hesss law to determine rH for the reaction Cgraphite O2g CO2g we ...View the full answer

Answered By

Issa Shikuku
I have vast experience of four years in academic and content writing with quality understanding of APA, MLA, Harvard and Chicago formats. I am a dedicated tutor willing to hep prepare outlines, drafts or find sources in every way possible. I strive to make sure my clients follow assignment instructions and meet the rubric criteria by undertaking extensive research to develop perfect drafts and outlines. I do this by ensuring that i am always punctual and deliver quality work.
5.00+
6+ Reviews
13+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Gebruik Hess se Wet om die reaksiewarmte vir die proses te bepaal. Use Hess's Law to determine the heat of reaction for the process. CH4(g) + 6HCl(g) 2CHCl3(g) + 4H(g) i. 2C(s) + 2H(g) CH4(g) | AH =...
-
The purpose of ERISA is to create transparency, accountability, and prevent the mismanagement of the investments made by participants in Plans. This week we are examining the requisite requirements...
-
Funtime, Inc., manufactures video game machines. Market saturation and technological innovations caused pricing pressures that resulted in declining profits. To stem the slide in profits until new...
-
A 16 lb weight is attached to a spring with a spring constant equal to 2 lb/ft. Neglect damping. The weight is released from rest at 3 ft below the equilibrium position. At t = 2r sec, it is struck...
-
For each of the following account balances for a manufacturing company, place a ( in the appropriate column indicating that it appears on the balance sheet, the income statement, the manufacturing...
-
Based on Websters research notes, which company would most likely be described as having high-quality cash fl ow? A . BIG Industrial B . Construction Supply C . Dynamic Production
-
Brian Rafferty ingested finasteride, a drug prescribed to him to treat an enlarged prostate. Finasteride is a generic version of Proscar, a drug manufactured by Merck. Some time after he started to...
-
Carmin Kovach is single and has two children from her previous marriage. Anika, 9, lives with Carmin. Julius, 11, lives with his father, Ray. Carmin pays alimony of $400 per month to Ray. The...
-
What are some business benefits of adopting the relational database model within an organization? How does a relational model create or enhance business value? Are there any limitations to using a...
-
Exhibit 13.6 presents selected hypothetical data from projected financial statements for Steak 'n Shake for Year +1 to Year +11. The amounts for Year +11 reflect a long-term growth assumption of 3%....
-
Use Hesss law to determine r H for the reaction C 3 H 4 (g) + 2 H 2 (g) C 3 H 8 (g), given that H(g) + O2(8) O(g) C3H4(g) + 4O2(g) C3H8(g) + 5O2(g) HO(1) A,H -285.8 kJ mol-1 3 CO2(g) + 2HO(1) A,H...
-
Write an equation to represent the combustion of thymol referred to in Exercise 44. Include in this equation the values for U and H. Exercise 44 A 1.397 g sample of thymol, C 10 H 14 O(s) (a...
-
Rad Co. provides the following sales forecast and production budget for the next four months: The company plans for finished goods inventory of 120 units at the end of June. In addition, each...
-
Supposean investment requires the company to increase inventories by $30,000 at the beginning of the project. However, the financial manager expects that accounts payable will increase by $10,000...
-
The following system has two loads. The first load Z draws an apparent power of 30 kVA with a power factor of 0.9 lagging. The second load draws 40 kW of power with a power factor of 0.795 lagging....
-
Daisey's Doughnuts and Gourmet Coffee is considering a new store location. The projected fixed annual cash operating costs for a store are $435,000 a year, variable costs are 52 percent of sales, and...
-
What is his gross income and adjusted gross income , assuming Phil Rupp files as head of household? During 2021, Phil Rupp presents the following transactions: Bank loan proceeds received (to...
-
Mr. Bala aged 30 is an accountant with an audit firm since five years ago. Recently he has made a booking for one unit of double storey terrace from a housing developer at a cost of RM300,000. Hehas...
-
Se9. Using the data in SE2, calculate the cost of ending inventory and cost of goods sold according to the LIFO method under the perpetual inventory system. In SE2, Assume the following data with...
-
In Problems, solve each system of equations. x + 2y + 3z = 5 y + 11z = 21 5y + 9z = 13
-
Change in PrincipleInventory Methods Whitman Company began operations on January 1, 2008, and uses the average cost method of pricing inventory. Management is contemplating a change in inventory...
-
Accounting Change Ramirez Co. decides at the beginning of 2010 to adopt the FIFO method of inventory valuation. Ramirez had used the LIFO method for financial reporting since its inception on January...
-
Accounting Change Linden Company started operations on January 1, 2006, and has used the FIFO method of inventory valuation since its inception. In 2012, it decides to switch to the average cost...
-
The figure gives the acceleration of a 5.0 kg particle as an applied force moves it from rest along an x axis from x=0 to x=9.0 m. The scale of the figure's vertical axis is set by a, 8.0 m/s. How...
-
4 . What is the duration of a four-year, $1,500 bond that pays a coupon (annual) of 12% that trades at a yield of 16%. Calculate is the expected change in the bonds price if interest rates fall by...
-
Your company has arranged a revolving credit agreement for up to $78 million at an interest rate of 1.47 percent per quarter. The agreement also requires your company to maintain a compensating...

Study smarter with the SolutionInn App


