What is the (a) Degree of ionization and (b) Percent ionization of propionic acid in a solution
Question:
What is the
(a) Degree of ionization and
(b) Percent ionization of propionic acid in a solution that is 0.45 M CH3CH2CO2H?
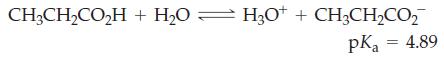
Transcribed Image Text:
CH3CH₂CO₂H + H₂O — H3O+ + CH3CH₂CO₂ pka = 4.89
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a Degree of ionization The degree of ionization is the fraction of the ini...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Exploring the degree of ionization equation in Study Question 127: Calculate the degree of ionization, , for formic acid at the following concentrations: 0.0100 M, 0.0200 M, 0.0400 M, 0.100 M, 0.200...
-
A handbook lists the following formula for the percent ionization of a weak acid. (a) Derive this equation. What assumptions must you make in this derivation? (b) Use the equation to determine the...
-
A 0.400-g sample of propionic acid was dissolved in water to give 50.0 mL of solution. This solution was titrated with 0.150 M NaOH. What was the pH of the solution when the equivalence point was...
-
Gray Corporation began operations on January 1, 2017. The following information is available for Gray Corporation on December 31, 2017. Prepare an income statement, a retained earnings statement, and...
-
What are stock purchase warrants? What are the similarities and key differences between the effects of warrants and those of convertibles on the firms capital structure and its ability to raise new...
-
Access has five choices on the Menu bar. One of them is Create. What are the other four?
-
Describe the role of an organizations personnel in compliance and antifraud efforts.
-
Ballard Co. reported $145,000 of net income for 2008. The accountant, in preparing the statement of cash flows, noted several items occurring during 2008 that might affect cash flows from operating...
-
There are four types of relational models: communal sharing, authority ranking, equality matching, and market pricing. Which relational models do you use in your interpersonal interactions? Is one...
-
What is the (a) Degree of ionization and (b) Percent ionization of ethylamine, C 2 H 5 NH 2 , in a 0.85 M aqueous solution?
-
In the diagram below, the sketch on the far left represents the [OH - ] present in an ammonia solution of molarity c. If the solution is diluted to half its original molarity, which of the sketches...
-
Using the information in P22-3A, compute the overhead controllable variance and the overhead volume variance. Data from P22-3A Standard (per unit) 8 yards at $430 per yard Cost Element Direct...
-
A Canadian LIC caters to mostly US-based customers and underwrites insurance policies in US dollars (USD). On the other hand, it maintains a portfolio of investments in Spain in euros (EUR). The...
-
Information: On January 1, 2015, the Herco Company purchased 70% of the outstanding voting share of the Wing Company for $ 850,000 in cash. On that date, the Wing Company had retained earnings of $...
-
D Question 3 For machines U and V. details are provided. What will be the Delta IRR and which machine should be selected? Initial cost Life in years Inflation (for costs and benefits) MARR Project...
-
1. The Salisbury Corporation is considering four mutually exclusive alternatives for a major capital investment project. All alternatives have a useful life of 10 years with no salvage value at the...
-
Is there any way to minimize capital gains if you have to sell shares less than 12 months? I. Calculate James's capital gains liability if he were to sell his Movonix Ltd Shares. Assume that James...
-
Country Products manufactures quilt racks. Pine is introduced in Department 1, where the raw material is cut and assembled. In Department 2, completed racks are stained and packaged for shipment...
-
B.) What is the approximate concentration of free Zn 2+ ion at equilibrium when 1.0010 -2 mol zinc nitrate is added to 1.00 L of a solution that is 1.080 M in OH - . For [Zn(OH) 4 ] 2- , K f = 4.610...
-
A company reports the following: Net income $120,000 Preferred dividends 20,000 Average stockholders equity 600,000 Average common stockholders equity 500,000 Determine (a) The rate earned on...
-
A company reports the following: Net income $ 180,000 Preferred dividends 12,000 Average stockholders equity 1,200,000 Average common stockholders equity 800,000 Determine (a) The rate earned on...
-
A company reports the following: Net income $340,000 Preferred dividends $40,000 Share of common stock outstanding 40,000 Market price per share of common stock $60.00 (a) Determine the companys...
-
Depreciation homework assignment Asset Date acquired Cost basis Computer 1/15/2019 1,500 Furniture 1/25/2019 5,000 Car 5/18/2019 12,000 Equipment 10/1/2019 19,000 Equipment 3/23/2020 10,000 Computer...
-
A wheel rotating with a constant angular acceleration turns through 10 revolutions dur- ing a 3 s time interval. Its angular velocity at the end of this interval is 12 rad/s. What is the angular...
-
A baseball is hit when it is 3 feet above the ground. It leaves the bat with initial 152ft speed of making an angle of 20 with the horizontal. At the instant the ball is sec hit, an instantaneous...

Study smarter with the SolutionInn App


