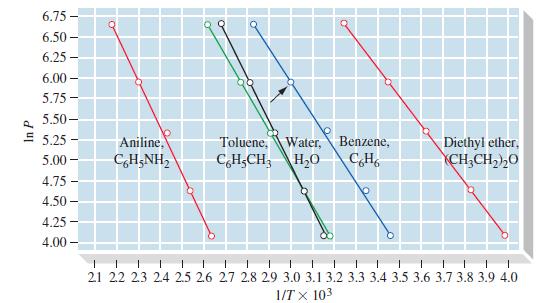
With reference to Figure 12-20, which is the more volatile liquid, benzene or toluene? At approximately what
Question:
With reference to Figure 12-20, which is the more volatile liquid, benzene or toluene? At approximately what temperature does the less volatile liquid have the same vapor pressure as the more volatile one at 65 °C?
Figure 12-20
Transcribed Image Text:
6.75 - 6.50- 6.25- 6.00- 5.75 - 5.50- 5.25 5.00- 4.75 - 4.50- 4.25- 4.00- Aniline, CHẠNH, O Toluene, Water, C6H-CH3 H₂O Benzene, C6H6 Diethyl ether, CH₂CH₂)20 1 1 1 I 1 I 1 1 1 I I 1 I 1 1 I I 1 1 21 22 23 24 25 26 27 28 29 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 3.9 4.0 1/TX 103
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The more volatile liquid in Figure 1220 is benzene Volatility is the tendency of a liquid to evap...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Selected information from Indigo Books & Music Inc.'s income statements for three recent years follows (dollars in thousands): Instructions (a) Calculate gross profit, and profit from operations, for...
-
Guarino and two others (plaintiffs) died of gas asphyxiation and five others were injured when they entered a sewer tunnel without masks to answer the cries for help of their crew leader, Rooney....
-
Use your knowledge of directing effects, along with the following data, to deduce the directions of the dipole moments in aniline andBromobenzene. Br- -NH2 Br- -NH2 H = 1.52 D H = 2.91 D A= 1.53 D
-
Redesign the fractionator of Example 6.8 for a reflux ratio that is twice the minimum. Determine the diameter of the tower, the height of packing in the stripping and rectifying sections, and the...
-
Gift Shop UBIT. A local exempt organization that trains at-risk youth for employment has an annual operating budget of $300,000, which includes revenue from operating a gift shop in a nearby hotel...
-
2. An engineer is planning for a 15-year retirement. In order to supplement his pension and offset the anticipated effects of inflation, he intends to withdraw $6000 at the end of the first year, and...
-
The vapor pressure of trichloromethane (chloroform) is 40.0 Torr at -7.1 C. Its enthalpy of vaporization is 29.2 kJ mol -1 . Calculate its normal boiling point.
-
The normal boiling point of acetone, an important laboratory and industrial solvent, is 56.2 C and its vap H is 25.5 kJ mol -1 . At what temperature does acetone have a vapor pressure of 375 mmHg?
-
a. Use the expansions of cos(2x + x) and cos(2x x) , to prove that: cos 3x + cos x + 2 cos 2x cos x b. Solve cos 3x + cos 2x + cos x > 0 for 0 < x < 360.
-
1. Explain on the financial statement analysis. 2. Explain on EIGHT (8) financial ratio. 3. Describe FIVE (5) types of financial objectives.
-
Like other direct-to-consumer startup companies such as Dollar Shave Club and Warby Parker, Glossier also offers its customers extremely low prices thanks to its direct-to-consumer business model....
-
One Day in the Seat of the Chair of the Federal Reserve Board. Imagine that you have been given the opportunity to serve as Chair of the US Federal Reserve Board. Please answer the following: 1)Your...
-
List all of the financial documents that you use in your department Select one of these documents and give a flow chart outlining how the documentis checked and processed Describe the personnel who...
-
Module 7- project 2 1. I created a new file in the PyCharm integrated development environment (IDE), title it "TextBasedGame.py. I cannot get the items in my dictionary to As you develop your code,...
-
On June 30, 2013, Papa Phil Inc. leased 200 pizza ovens for its chain of restaurants from Pizza Inc. The lease agreement calls for Papa Phil to make semiannual lease payments of $562,907 over a...
-
Element compound homogeneous mixture (heterogeneous mixture) 4) A piece of gold has a mass of 49.75 g. What should the volume be if it is pure gold? Gold has a density of 19.3 g/cm (3 points) D=m/v...
-
Zero Coupon Bonds suppose your company needs to raise $20 million and you want to issue 30-year bonds for this purpose. Assume the required return on your bond issue will be 7 percent, and youre...
-
Finding the Maturity youve just found a 10 percent coupon bond on the market that sells for par value. What is the maturity on this bond?
-
Real Cash Flows you want to have $1 million in real dollars in an account when you retire in 40 years. The nominal return on your investment is 11 percent and the inflation rate is 4.5 percent. What...
-
If total assets decreased by $88,000 during a period of time and stockholders' equity increased by $71,000 during the same period, then the amount and direction (increase or decrease) of the period's...
-
Bunnell Corporation is a manufacturer that uses job-order costing. On January 1, the company's inventory balances were as follows: Raw materials $ 84,500 15 Work in process Finished goods $ 35,000 $...
-
On December 31, Jarden Company's Allowance for Doubtful Accounts has an unadjusted credit balance of $16,000. Jarden prepares a schedule of its December 31 accounts receivable by age Accounts...

Study smarter with the SolutionInn App


