Determine the shapes of each of the following molecules and then, using the data in Table 2.2,
Question:
Determine the shapes of each of the following molecules and then, using the data in Table 2.2, state whether each is expected to be polar or not:
(a) H2S;
(b) CO2;
(c) SO2;
(d) BF3;
(e) PF5 ;
(f) cis-N2F2;
(g) trans-N2F2;
(h) HCN.
Table 2.2
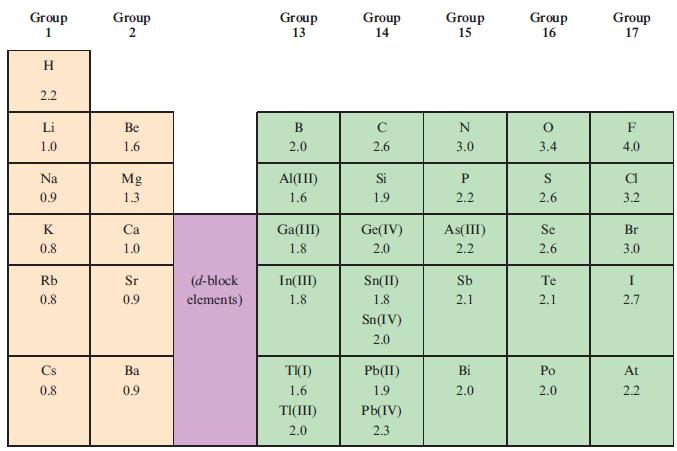
Transcribed Image Text:
Group 1 I H 2.2 Li 1.0 Na 0.9 K 0.8 Rb 0.8 Cs 0.8 Group 2 Be 1.6 Mg 1.3 Ca 1.0 Sr 0.9 Ba 0.9 (d-block elements) Group 13 B 2.0 Al(III) 1.6 Ga(III) 1.8 In(III) 1.8 TI(I) 1.6 TI(III) 2.0 Group 14 с 2.6 Si 1.9 Ge(IV) 2.0 Sn(II) 1.8 Sn(IV) 2.0 Pb(II) 1.9 Pb(IV) 2.3 Group 15 N 3.0 P 2.2 As(III) 2.2 Sb 2.1 Bi 2.0 Group 16 O 3.4 S 2.6 Se 2.6 Te 2.1 Po 2.0 Group 17 F 4.0 CI 3.2 Br 3.0 I 2.7 At 2.2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
The number of electrons in the valence shell of a central p block atom tends to control the shape of molecules Polarity is a property of molecules The net molecular dipole moment of a polyatomic speci...View the full answer

Answered By

User l_1013947
I possess a comprehensive understanding of programming languages such as C++, Python, HTML, CSS, and Jupyter Notebook. These technical skills enable me to develop robust software solutions and create visually appealing web pages. With my expertise in coding, I can effectively tackle complex programming tasks and deliver high-quality results.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
The board of directors of Portand Ltd are considering two mutually exclusive investments each of which is expected to have a life of five years. The company does not have the physical capacity to...
-
Using the data in the following table, predict the sign and magnitude of ÎH° for each of the following reactions. In each case, identify whether the reaction is expected to be endothermic...
-
Which of the following is most likely to be recognized as a defining characteristic of the public good? a) The good is a non-rival element. B) the good is a non-excludable item C) The good is both A...
-
Avery Alton practices law under the business title Avery Alton, Attorney at Law, Inc. During September, her law practice engaged in the following transactions: Sep 1 Sold $50,000 of common stock to...
-
Imagine 100 book bags, each of which contains 1,000 poker chips. Forty-five bags contain 700 black chips and 300 red chips. The other 55 bags contain 300 black chips and 700 red chips. You cannot see...
-
A construction contract has the following language: It is the responsibility of the contractor to inspect and become familiar with the Project and to acquaint itself thoroughly with all conditions...
-
Materials and manufacturing labor variances, standard costs. Dunn, Inc. is a privately held furniture manufacturer. For August 2009, Dunn had the following standards for one of its products, a wicker...
-
Two large conducting plates on insulating stands are placed a distance D = 50 cm apart, as shown at right. The inner surface of one has a charge density of +0; the other, - The charge density on the...
-
In the following table, match a species in list 1 with an isoelectronic partner in list 2. Some species may have more than one partner. Qualify how you have interpreted the term isoelectronic. List 1...
-
Draw Lewis structures for (a) CO 2 , (b) SO 2 , (c) OF 2 (d) H 2 CO.
-
A block on a frictionless table is connected as shown in FIGURE P15.74 to two springs having spring constants k 1 and k 2 . Show that the blocks oscillation frequency is given by f = f 1 2 + f 2 2...
-
The president has just signed a new budget that drastically cuts taxes without decreasing government spending. When asked about the dramatic increase in deficits that will occur due to the tax cuts,...
-
Predict how the Fed would likely respond if the output gap became more positive, so that output moved from being 0.1% above potential output to being 3% above potential output, and inflation rose...
-
Determine if the following changes to the economy are examples of financial, spending, or supply shocks. For each case, explain whether the IS, MP, or Phillips curve will shift, and in what...
-
Discuss the advantages and disadvantages of ratio analysis. Montgomery Organizations plc, whose annual accounts are given below. The firm has 14,685,856 shares and the share price is 2.78. Fixed...
-
Calculating Future Values Calculate the future value of a 100 cash flow for the following combinations of rates and times: (a) r = 8%; t = 10 years (b) r = 8%; t = 20 years (c) r = 4%; t = 10 years...
-
Explain what it means if a loan requires blended payments.
-
In muscle tissue, the ratio of phosphorylase a to phosphorylase b determines the rate of conversion of glycogen to glucose 1phosphate. Classify how each event affects the rate of glycogen breakdown...
-
The following Latimer diagrams show the standard reduction potentials E /V for some oxidation states of iron in acid and alkaline solution: (a) Plot a Frost diagram showing the states of Fe under...
-
Data shown below refer to redox couples for the Group 8 elements Fe and Ru. (a) Comment on the relative stability of Fe 2+ and Ru 2+ in acidic aqueous solution. (b) Give a balanced equation for the...
-
Many of the tabulated data for standard potentials have been determined from thermochemical data rather than direct electrochemical measurements of cell potentials. Carry out a calculation to...
-
On January 1, Ford Engineering and Consultancy signs a contract to build a hotel. The construction is scheduled to begin on January 5, 2021 and is estimated to take four years to complete. The...
-
Both Austin and Jasmine purchased items on account thru Mason Landscaping. Austin will pay its account within 30 days. Jasmine signed a written promise to pay within 90 days with a 1% monthly...
-
Assume there are 10 equally sized hospitals in a market area. 1. Calculate the HHI. 2. Consider the case where two hospitals in the market area inform the Department of Justice (DOJ) that they wish...

Study smarter with the SolutionInn App


