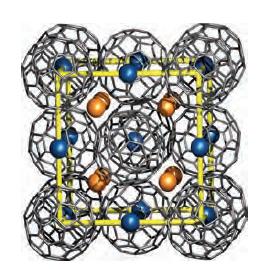
Figure 14.10 shows a unit cell of K 3 C 60 . From the structural information given,
Question:
Figure 14.10 shows a unit cell of K3C60. From the structural information given, confirm the stoichiometry of this fulleride.
Figure 14.10.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The stoichiometry of K3C60 can be confirmed from the structural information given in Figure 1410 as ...View the full answer

Answered By

Moses mwangi
With prior writing experience, be sure that I will give a great grade, If not an A+, it will be something close to this. My reviews speaks it all, Try me!!
4.80+
78+ Reviews
157+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Figure 15.23 shows a unit cell of FeSb 2 O 6 . (a) How is this unit cell related to the rutile-type structure? (b) Why can the solid state structure of FeSb 2 O 6 not be described in terms of a...
-
MgF 2 has a TiO 2 -type structure. (a) Sketch a unit cell of MgF 2 , (b) Confirm the stoichiometry of MgF 2 using the solid state structure.
-
(a) Na 3 N remained an elusive compound until 2002. Calculate a value for f H o (Na 3 N, s) using data from Appendices 8 and 10, and the following estimated values of H(298 K): Comment on whether...
-
Which of the following is not true regarding a promissory note? a. Promissory notes may not be transferred to another party by endorsement. b. Promissory notes may be sold to another party. c....
-
You are employed by a business consulting firm as an information systems specialist. You have just begun an assignment with a startup company and are discussing with the owner her need for an...
-
In problem, determine whether each relation represents a function. For each function, state the domain and range. Birthday Person Jan. 8 Elvis Colleen Kaleigh Mar. 15 Sept. 17 Marissa
-
Consider the following cash flow profile, and assume MARR is 10 percent/year and the finance rate is 4 percent/year. a. Determine the MIRR for this project. b. Is this project economically...
-
Northstar Company uses ABC to account for its chrome wheel manufacturing process. Company managers have identified four manufacturing activities that incur manufacturing overhead costs: materials...
-
List and describe/discuss the five advantages of intermediate sanctions over traditional probation or incarceration.
-
Comment on each of the following observations. (a) The carbides Mg 2 C 3 and CaC 2 liberate propyne and ethyne respectively when treated with water, reaction between ThC 2 and water produces mixtures...
-
Discuss the trends in data in Table 12.4. Data from Table 12.4. Metal, M Mg Ca Sr Ba MF2 -1113 -1214 -1213 -1200 AH/kJ mol MC1 -642 -795 -828 -860 MBr -517 -674 -715 -754 MI -360 -535 -567 -602
-
Which electrons are most responsible for the properties of an atom?
-
On January 10 of the current year, Vicky transfers to Loretta Corporation a machine purchased three years ago for $97,000. On the transfer date, the machine has a $64,000 adjusted basis and a...
-
QUESTION 4 What is the purpose of the System.out.print method in Java? O 1. Reads a line from the console O2. Prints a string to the console without a newline 3. Prints a line to the console O 4....
-
[15] Which register transfer statement is equivalent to WRITE microopeation: a) DRAC b) DR M[AR] c) M[AR] DR d) AC DR
-
K Does margin buying involve borrowing money from your broker? A. Yes B. No
-
[16] Mapping the opcode 0110 to a 7 bit address will give: hi obo a) 0001100 b) 0011000 c) 0000110 d) 0000011 115
-
When a least squares line is fit to the 8 observations in the natural gas consumption data, we obtain SSE = 2.568. Calculate s2 and s.
-
Which of the following is NOT a magnetic dipole when viewed from far away? a) A permanent bar magnet. b) Several circular loops of wire closely stacked together with the same current running in each...
-
Figure 26.68 shows Mssbauer spectra of a sample of ferredoxin from chloroplasts at 77 K. Interpret the data with regard to the oxidation states an spin states of the two Fe atoms and comment on the...
-
J.A. Botas et al. discuss the catalytic conversion of vegetable oils into hydrocarbons suitable for use as biofuels (Catal. Today, 2012, 195, 59). What are the most important features of catalysts...
-
Apart from direct O-atom transfer (Fig. 26.50), another mechanism proposed for Mo enzymes is indirect O-atom transfer, also known as coupled electron-proton transfer. In this mechanism, shown in Fig....
-
Learned some of the most important concepts in the course ACC-421-Q3329 Auditing and Forensic Acct. Two of them are the concepts of expert witness and fraud investigator in forensic accounting. This...
-
1. Who is liable for the federal and state income taxes on the $523,000? a. The cooperative's patrons b. The cooperative c. Both the cooperative and the cooperative's patrons 2) If the marginal tax...
-
Harry and Megan, two unrelated individuals, are considering forming the Royal Fashion Shoe Corporation in the beginning of 2022. Harry plans to transfer the following assets to the new corporation in...

Study smarter with the SolutionInn App


