How would you attempt to (a) Estimate the crystal field stabilization energy of FeF 2 , (b)
Question:
How would you attempt to
(a) Estimate the crystal field stabilization energy of FeF2,
(b) Determine the overall stability constant of [Co(NH3)6]3+ in aqueous solution given that the overall formation constant for [Co(NH3)6]2+ is 105, and:
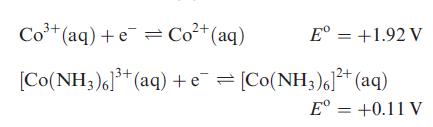
Transcribed Image Text:
Co+(aq)+e=Cot(aq) Eº = +1.92 V [Co(NH3)6]³+ (aq) + e¯ = [Co(NH3)6]²+ (aq) Eº = +0.11 V
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
a Estimating the crystal field stabilization energy CFSE of FeF2 Crystal field stabilization energy is a term used in coordination chemistry to descri...View the full answer

Answered By

Surendar Kumaradevan
I have worked with both teachers and students to offer specialized help with everything from grammar and vocabulary to challenging problem-solving in a range of academic disciplines. For each student's specific needs, I can offer explanations, examples, and practice tasks that will help them better understand complex ideas and develop their skills.
I employ a range of techniques and resources in my engaged, interesting tutoring sessions to keep students motivated and on task. I have the tools necessary to offer students the support and direction they require in order to achieve, whether they need assistance with their homework, test preparation, or simply want to hone their skills in a particular subject area.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How would you attempt to assess whether the technology used by an industry in a developing country was inappropriate?
-
You have heard that the average American male is 69.7 inches tall. Yet almost all of the adult males in your personal world seem to be between 5 and 6 feet tall, with a few just over 6 feet. So how...
-
The recently completed new building to house the exhibits and staff of the Central City Museum was located adjacent to the campus of a private university. The new building was financed by the...
-
Mary, Ali and Nzioka are trading as Mali enterprises. They share profits and losses in the ratio of 2 = 1 1 respectively. The following is a statement of comprehensive income for the year ended 31st...
-
A construction project is broken down into the following 10 activities: a. Draw the network diagram. b. Find the critical path. c. If activities 1 and 10 cannot be shortened, but activities 2 and 9...
-
In the following Verilog process A, B, C, and D are all registers that have a value of 0 at time = 10ns. If E changes from 0 to 1 at time = 20ns, specify the time(s) at which each signal will change...
-
Mrs. Clarks Foods was an Iowa company engaged in the business of distributing juice beverages. International Suntrade and Miller & Smith Foods were Canadian companies that acted as brokers...
-
(2-Year Worksheet) On January 1, 2010, Cunningham Company has the following defined benefit pension plan balances.? Projected benefit obligation ? ? ? ? ? ? ? ? ? ? ? ? ? ? ? ? ? ? ? ?$4,500,000 Fair...
-
Consider the following ridiculously simple linear system: 10 (9)(2)-(0) a. What is the solution of this linear system? You don't have to show your work. b. Set up a Neumann iteration with initial...
-
Figure 21.44 shows the change in concentration of [MnO 4 ] with time during a reaction with acidified oxalate ions. (a) Suggest a method of monitoring the reaction. (b) Explain the shape of the...
-
Suggest the formula and structure of the mononuclear complex formed between Cr 3+ and ligand 21.83. Comment on possible isomerism. CO N CO N (21.83) CO
-
What is the advantage of loading a counter synchronously?
-
The two masses P (5 Kg) and Q (unknown) are suspended as shown by this network of cords. The points A and D correspond to the attachments to the walls. www What should be the mass Q so that this...
-
Compute the monthly payments on a 3-year lease for a $26,484 car if the annual rate of depreciation is 13% and the lease's annual interest rate is 5.3%. Round your answer to the nearest dollar.
-
If a 50 N block is resting on a steel table with a coefficient of static friction us = 0.74, then what minimum force is required to move the block?
-
A wholesale merchandising company uses the FIFO cost flow assumption. The beginning inventory balance was zero. Below is a worksheet showing inventory transactions for December 2020. What is the...
-
Consider the equal and opposite charges shown below. YA +Q++ a a (a) Show that at all points on the x-axis for which |x| a, Ex Browse... No file selected. This answer has not been graded yet....
-
Paul and Donna Decker are married taxpayers, ages 44 and 42, respectively, who file a joint return for 2014. The Deckers live at 1121 College Avenue, Carmel, IN 46032. Paul is an assistant manager at...
-
Which of the following raises the credibility of areport? Which of the following raises the credibility of a report? Multiple Choice avoiding predictions avoiding the use of cause-effect statements...
-
Predict the product(s) for each of the following reactions. In each case, make sure to consider the number of chirality centers being formed. a. b. c. d. e. f. Os0, (catalytic) NMO 1) OsO, 2) NaHSO,...
-
Draw the propagation steps that achieve the autooxidation of diethyl ether to form a hydroperoxide: OOH A hydroperoxide Diethyl ether
-
Myo-Inositol is a polyol (a compound containing many OH groups) that serves as the structural basis for a number of secondary messengers in eukaryotic cells. Draw the more stable chair conformation...
-
Policy gets updated annually in most healthcare organizations. This is due to several reasons. Policy is a course or principle of action adopted or proposed by a government, party, business, or...
-
Think about how policy affects every aspect of what an employee does at work. Policy is often difficult to change. The reason is that policy refers to the laws that healthcare professionals live by...
-
A merchandising company has the following inventory information available for May: Number of Units Cost per unit Date Description May 1 Beginning Inventory 50 $20 May 19 Purchases 75 $25 The company...

Study smarter with the SolutionInn App


