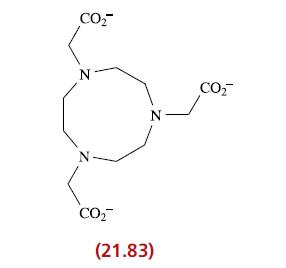
Suggest the formula and structure of the mononuclear complex formed between Cr 3+ and ligand 21.83. Comment
Question:
Suggest the formula and structure of the mononuclear complex formed between Cr3+ and ligand 21.83. Comment on possible isomerism.
Transcribed Image Text:
CO₂ N CO₂ N (21.83) CO₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The ligand 2183 appears to be described as CONCONCO based on the provided structure This ligand can ...View the full answer

Answered By

Chandrasekhar Karri
I have tutored students in accounting at the high school and college levels. I have developed strong teaching methods, which allow me to effectively explain complex accounting concepts to students. Additionally, I am committed to helping students reach their academic goals and providing them with the necessary tools to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) A 6-coordinate complex may be obtained by crystallizing anhydrous CaI 2 from THF solution at 253 K. In contrast, when anhydrous BaI 2 is crystallized from THF at 253 K, a 7-coordinate complex is...
-
Treatment of an aqueous solution of NiCl 2 with H 2 NCHPhCHPhNH 2 gives a blue complex ( eff = 3:30 B ) which loses H 2 O on heating to form a yellow, diamagnetic compound. Suggest explanations for...
-
Reduction of Ir 4 (CO) 12 with Na in THF yields the salt Na[Ir(CO) x ] (A) which has a strong absorption in its IR spectrum (THF solution) at 1892 cm 1 . Reduction of A with Na in liquid NH 3 ,...
-
The net present value and internal rate of return desirability measures for two mutually exclusive investments being considered by Stockton Corporation to follow. Year NPV IRR R 161 14.60% S 138...
-
The following table represents a plan for a project: a. Construct the appropriate network diagram. b. Indicate the critical path. c. What is the expected completion time for the project? d. You can...
-
(a) Assume D 1 = 0, D 2 = 5, and D 1 changes to 1 at time = 10ns. What are the values of D 1 and D 2 after the following code has been executed once? Do the values of D 1 and D 2 swap? always @ (D1)...
-
Police in Albemarle County, Virginia, were on the lookout for a stolen orange and black motorcycle that had eluded them in two previous traffic incidents. Officer David Rhodes drove past the home of...
-
Haas Company manufactures and sells one product. The following information pertains to each of the companys first three years of operations: Variable costs per unit: Manufacturing: Direct materials ....
-
Define the relational model? What does the relational model consist of? What are the 4 relational models?Discuss
-
How would you attempt to (a) Estimate the crystal field stabilization energy of FeF 2 , (b) Determine the overall stability constant of [Co(NH 3 ) 6 ] 3+ in aqueous solution given that the overall...
-
(a) Which of the following complexes would you expect to suffer from a JahnTeller distortion: [CrI 6 ] 4 , [Cr(CN) 6 ] 4 , [CoF 6 ] 3 and [Mn(ox) 3 ] 3 ? Give reasons for your answers. (b) [Et 4 N] 2...
-
You are presented with the following information relating to Messiter plc: Required: (a) Using the historical cost financial statements and stating the formulae you use, calculate the following...
-
A small company that trains executives on how to act when working in Japan had $70,000 worth of inventory on January 1, 2018, and it had $50,000 worth of inventory on December 31, 2018. If its cost...
-
A lawsuit must at least have a clear object or a lawsuit must fulfill the elements of a lawsuit such as having the identity of the plaintiff and defendant. If it does not fulfill the requirements or...
-
ULA V-(0.750 m/s)i A cruise ship with a mass of 107 kg strikes a pier at a speed of 0.750 m/s. After traveling 5.93 meters, damaging the ship and the pler, determine the average force in Newtons...
-
Describe the seven-step process of managing anger introduced by Dr Lynn McClure in detail.
-
Find all the zeros of the function given the zeros already provided. 6) f(x) = 5x + 4x - 11x + 2; -2 5 7) f(x)= 9x - 36x + 17x + 30; 3
-
Alice J. and Bruce M. Byrd are married taxpayers who file a joint return. Their Social Security numbers are 123-45-6789 and 111-11-1111, respectively. Alice's birthday is September 21, 1966, and...
-
Which of the following is FALSE regarding the purchasing power parity (PPP). a. The PPP is a manifestation of the law of one price b. The PPP says that a country with a higher expected inflation can...
-
Each of the following molecules has one plane of symmetry. Find the plane of symmetry in each case: a. b. c. d. e. f. CI Me Me Br
-
Compound A has molecular formula C5H12, and monobromination of compound A produces only compound B. When compound B is treated with a strong base, a mixture is obtained containing compound C and...
-
Compare the Lewis dot structure of nitrogen and phosphorus and explain why you might expect these two atoms to exhibit similar bonding properties?
-
2. Consider the following graph and write a java program that will: 4 (b 10 8 7 6 a 9 5 9 8 2 2 C f 1 a. Generate a minimum spanning tree using Prim's algorithm and finally compute the total cost b....
-
1. Write a java program named BSTree.java that will: a. Generate 100 random integer numbers ranging from 1-99. b. Build a Binary Search Tree using this set of numbers. c. Delete 50 and 60 from the...
-
A field party is capable of making taping observations with a standard deviation of 0.010 ft per 100-ft tape length. What standard deviation would be expected in a distance of 200 ft taped by this...

Study smarter with the SolutionInn App


