In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. The 19 F
Question:
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed.
The 19F NMR spectrum of the octahedral ion [PF5Me]‾ shows two signals (δ – 45.8 and –57.6 ppm). Why are two signals observed? From these signals, three coupling constants can be measured: JPF = 829 Hz, JPF = 680 Hz and JFF = 35 Hz. Explain the origins of these coupling constants.
Table 4.3
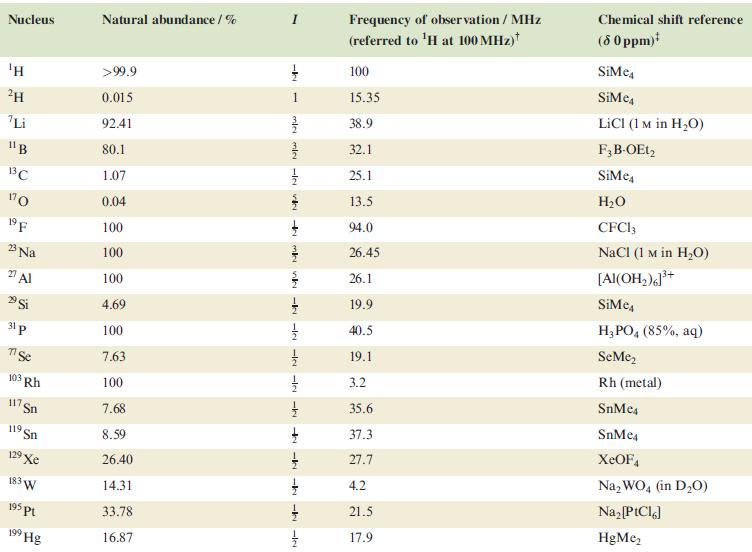
Transcribed Image Text:
Nucleus ΤΗ ²H Li 11B 13C 170 19 F 23 Na 2 Al 29 Si 31 p 7 Se 103 Rh 117. Sn 119 Sn 19 Xe 183 W 195 pt 199 Hg Natural abundance/% >99.9 0.015 92.41 80.1 1.07 0.04 100 100 100 4.69 100 7.63 100 7.68 8.59 26.40 14.31 33.78 16.87 miele v + mk nh -k -le -le-le - -ki -k -k -IN Frequency of observation / MHz (referred to ¹H at 100 MHz) 100 15.35 38.9 32.1 25.1 13.5 94.0 26.45 26.1 19.9 40.5 19.1 3.2 35.6 37.3 27.7 4.2 21.5 17.9 Chemical shift reference (8 0 ppm)* SiMe SiMe4 LICI (1 M in H₂O) F3B-OEt2 SiMe H₂O CFC13 NaCl (1 M in H₂O) [Al(OH₂)]³+ SiMc4 H3PO4 (85%, aq) SeMe₂ Rh (metal) SnMc4 SnMe4 XeOF4 Na₂WO4 (in D₂O) Na₂ [PtCl6] HgMe₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
The presence of two signals in the 19F NMR spectrum of the octahedral ion PF5Me is due to the inequivalent fluorine nuclei in the molecule In octahedr...View the full answer

Answered By

Akash M Rathod
I have been utilized by educators and students alike to provide individualized assistance with everything from grammar and vocabulary to complex problem-solving in various academic subjects. I can provide explanations, examples, and practice exercises tailored to each student's individual needs, helping them to grasp difficult concepts and improve their skills.
My tutoring sessions are interactive and engaging, utilizing a variety of tools and resources to keep learners motivated and focused. Whether a student needs help with homework, test preparation, or simply wants to improve their skills in a particular subject area, I am equipped to provide the support and guidance they need to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
19F is the only isotope of fluorine that occurs naturally, and it has a nuclear spin of + (a) Into how many peaks will the proton signal in the 1H NMR spectrum of methyl fluoride be split? (b) Into...
-
19F is the only isotope of fluorine that occurs naturally, and it has a nuclear spin of 1/2 (a) Into how many peaks will the proton signal in the 1H NMR spectrum of methyl fluoride be split? (b) Into...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. (a) In the 1 H NMR spectrum of compound 4.21, there is a triplet at 3.60 ppm (J 10.4 Hz). Assign the signal and...
-
The following are selected 2023 transactions of Ayayai Corporation. Sept. 1 Oct. 1 1 Purchased inventory from Indigo Ltd. on account for $47,200. Ayayai uses a periodic inventory system. Issued a...
-
Place the corresponding letter of the definition next to the term. _____ 1. Cost principle _____ 2. Business entity principle _____ 3. Generally accepted accounting principles _____ 4. Reliability...
-
Suggest a method to determine whether the TART retroposon is situated at the telomeres of each of the chromosomes in the Drosophilagenome.
-
Consider the calculation of an external rate of return (ERR). The positive cash flows in the cash flow profile are moved forward to \(t=n\) using what value of \(i\) in the \((F \mid P i, n-t)\)...
-
Using the payback period and unadjusted rate of return to evaluate alternative investment opportunities Louis Gallo owns a small retail ice cream parlor. He is considering expanding the business and...
-
Does the monopoly power wielded by professional sports leagues hurt the fans, players, citizens, local economies, or the sport? What solutions could be used ?
-
(a) If Na has the ground state electronic configuration of [Ne]3s 1 , why is NaCl EPR silent? (b) Sketch an EPR spectrum for an isotropic system in which an electron interacts with a 14 N (I = 1)...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. The 31 P{ 1 H} NMR spectrum of a CDCl 3 solution of the square planar rhodium(I) complex 4.22 exhibits a doublet of...
-
What factors determine a central bank's independence? What are the benefits of having an independent central bank?
-
For each variable cost per unit listed below, determine the total variable cost when units produced and sold are 100, 150, and 200 units. Direct materials Direct labor Variable overhead Selling...
-
Explain the term standard.
-
List three items that are required in order to prepare the budgeted income statement.
-
Calculate the unit product cost using variable costing. Round your answer to the nearest cent. Matthew Company had the following costs: Units produced Direct materials Direct labor Variable...
-
How is the cost of goods sold calculated when preparing a budget?
-
In August 2016, Heavy-lift Helicopters Ltd. purchased a new helicopter for its logging and heavy lift operations at a cost of $1.2 million. Based on past experience, management has determined that...
-
5. Convert the following ERD to a relational model. SEATING RTABLE Seating ID Nbr of Guests Start TimeDate End TimeDate RTable Nbr RTable Nbr of Seats RTable Rating Uses EMPLOYEE Employee ID Emp...
-
The potential of organofluoro compounds in materials chemistry is discussed in a paper by R. Berger and co-workers (Chem. Soc. Rev., 2011, 40, 3496). One group of compounds discussed is the...
-
Sketch a chloralkali cell. Show the half-cell reactions and indicate the direction of diffusion of the ions. Give the chemical equation for the unwanted reaction that would occur if OH migrated...
-
In their paper Recent discoveries of polyhalogen anions from bromine to fluorine (Z. Anorg. Allg. Chem., 2014, 640, 7, 1281), H. Haller and S. Riedel described the synthesis and structure elucidation...
-
The profile of the cables on a suspension bridge may be modeled by a parabola. The central span of the bridge is 1250 m long and 145 m high. The parabola y = 0.00037x gives a good fit to the shape of...
-
4) [9] Let A = {-3,-2,-1,0,1} and B = {-8,-6,0,6,10}. Define g: A B by g(x) = 2x - 8. a) What is the codomain of g? b) What is the range of g? c) What is the domain of g? d) Is g injective? Explain...
-
Write these decimals as fractions in lowest terms, and determine the prime factorization of the denominator in each case. (a) 3.28 (b) 0.18 (a) Write 3.28 as a fraction in lowest terms. 3.28 = 82 25...

Study smarter with the SolutionInn App


