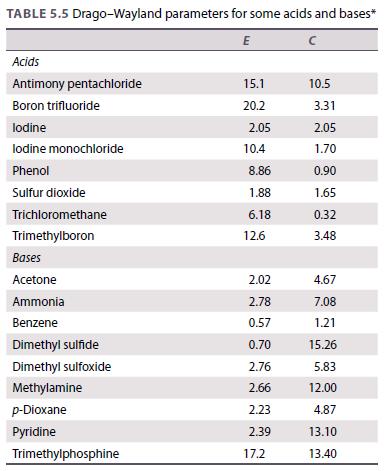
Use the data in Table 5.5 to calculate the enthalpy change for the reaction of iodine with
Question:
Use the data in Table 5.5 to calculate the enthalpy change for the reaction of iodine with phenol.
Table 5.5.
Transcribed Image Text:
TABLE 5.5 Drago-Wayland parameters for some acids and bases* E Acids Antimony pentachloride Boron trifluoride lodine lodine monochloride Phenol Sulfur dioxide Trichloromethane Trimethylboron Bases Acetone Ammonia Benzene Dimethyl sulfide Dimethyl sulfoxide Methylamine p-Dioxane Pyridine Trimethylphosphine 15.1 20.2 2.05 10.4 8.86 1.88 6.18 12.6 2.02 2.78 0.57 0.70 2.76 2.66 2.23 2.39 17.2 10.5 3.31 2.05 1.70 0.90 1.65 0.32 3.48 4.67 7.08 1.21 15.26 5.83 12.00 4.87 13.10 13.40
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
To calculate the enthalpy change for the reaction of iodine with phenol we can use the st...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
In the following exercises, you will use the data in the TAL Distributors database shown in Figure 2-1 in Chapter 2. (If you use a computer to complete these exercises, use a copy of the original TAL...
-
In the following exercises, you will use the data in the Solmaris Condominium Group database shown in Figures 1-21 through 1-25. (If you use a computer to complete these exercises, use a copy of the...
-
In the following exercises, you will use the data in the Solmaris Condominium Group database shown in Figures 1-21 through 1-25 in Chapter 1. (If you use a computer to complete these exercises, use a...
-
Three disease-carrying organisms decay exponentially in lake water according to the following model: Estimate the initial population of each organism (A, B, and C) given the followingmeasurements:...
-
Amner Manufacturing Company had an excellent year. The company hired a new marketing director in January. The new directors great motivational appeal inspired the sales staff, and, as a result, sales...
-
What is the purpose of the liability clause?
-
William Stem filed an action against Gary Braden, seeking to rescind a contract for the sale of an automobile and to obtain the return of the purchase price plus interest. The trial court granted...
-
Peninsula Candy Company makes three types of candy bars: Chewy, Chunky, and Choco-Lite (Lite). Sales volume for the annual budget is determined by estimating the total market volume for candy bars...
-
Pinehurst Company was formed in Year 1 and experienced the following accounting events during the year: 1. Issued common stock for $18,400 cash. 2. Earned cash revenue of $26,100. 3. Paid cash...
-
Identify the products from the reaction between the following pairs of reagents. In each case identify the species which are acting as a Lewis acid or a Lewis base in the reactions. (a) CsF + BrF 3...
-
For each of the following processes, identify the acids and bases involved and characterize the process as complex formation or acidbase displacement. Identify the species that exhibit Brnsted...
-
After 23 days, a 10-milligram sample of a radioactive material decays to 5 milligrams. After how many days will there be 1 milligram of the material?
-
How does the use of allusions to literary predecessors and canonical texts contribute to the development of literary traditions and genres, creating a dialogic relationship between past and present...
-
a. Project A costs $8,000 and will generate annual after-tax net cash inflows of $3,250 for 5 years. What is the payback period for this investment under the assumption that the cash inflows occur...
-
The Shape interface provides public getColor and setColor methods. The colors are defined in a public enum, in the Shape class, whose values include TRANSPARENT, BLACK, RED, ORANGE, YELLOW, GREEN,...
-
A wood crate sits in the back of a pickup truck. The coefficients of friction between the crate and the pickup truck are =0.61 and x=0.37. The pickup truck starts moving up a 11 slope. What is the...
-
On January 1, 2016, the Winderl Mining Co. paid $50 million for a copper mine owned by the state of Wyoming. To obtain the mine, Winderl agreed to restore the land to a suitable condition for other...
-
Maleic acid is an organic compound composed of 41.39% C, 3.47% H, and the rest oxygen. If 0.129 mole of maleic acid has a mass of 15.0 g, what are the empirical and molecular formulas of maleic acid?
-
If a process has a six-sigma capability, what is the process capability index? a. 1 b. 2 c. 6 d. 12
-
Using data from Appendix 11, and the value for the standard Gibbs energy of formation for PbS of 99 kJ mol 1 , determine a value for K sp for this salt. Data from Appendix 11 The concentration of...
-
In hydrochloric acid, HOI reacts to give [ICl 2 ] . Use the potential diagrams below to explain why HOI disproportionates in aqueous acidic solution, but does not when the acid is aqueous HCl. [03]...
-
(a) Using the potential diagram below (at pH 14), calculate E O3 2 /O 2 (b) Comment on the following data: (c) How valid is Fig. 8.4a for aqueous solutions at pH 2? Figure 8.4a. 03 +0.66 03- +1.25 E 0
-
You have decided to buy a 3,250 square-feet house in Denton, close to a good public school. The average price in the area is $190 per square feet. The bank will finance 90% of the house value, so you...
-
Consumer loans can be arranged in a similar fashion if the lender collects a share of future income instead of interest payments. For example, some people are eligible to arrange for income-based...
-
How do firms maximize profit? describes positive statements in economics. Offer some positive economic statements regarding profit maximization in business. describes consumer and producer surplus....

Study smarter with the SolutionInn App


