Careful measurements of the mass of a hydrogen-3 nucleus (tritium) yield a value of 3.01605 u. Compute
Question:
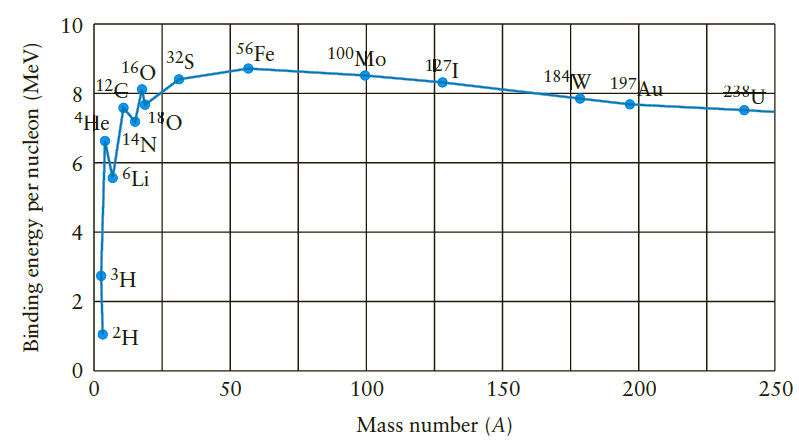
Careful measurements of the mass of a hydrogen-3 nucleus (tritium) yield a value of 3.01605 u. Compute the binding energy in MeV and the binding energy per nucleon for this radioactive nuclide. Compare your value with that for 3H given in Figure 11.16.
Transcribed Image Text:
10 100MO 56F. 1271 325 184W 197AU 160 126 180 238 4He 14N 6Li 4 ЗН 2 2H 250 200 150 100 50 Mass number (A) Binding energy per nucleon (MeV)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
The measured mass of a hydrogen3 nucleus tritium is given as 301605 u Thus we ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
Recession of the Moon Careful measurements of the earth-moon separation indicate that our satellite is presently moving away from us at approximately 3.0 cm per year. Neglect any angular momentum...
-
Define the unit dalton. From this definition, compute the mass of 1 Da in grams. The mean of 60 measurements of the mass of individual E. coli cells vaporized by MALDI and measured with a quadrupole...
-
The naturally occurring radioisotopes uranium-238 and uranium-235 have decay chains that end with the stable isotopes lead-206 and lead-207, respectively. Natural minerals such as zircons contain...
-
Why would senior managers want to distort factor prices by using cost allocations?
-
Refer to the previous exercise. a. What is the expected number of calls among the 25 that involve a fax message? b. What is the standard deviation of the number among the 25 calls that involve a fax...
-
At a price of $1.88 per pound, the supply for cherries in a large city is 16,000 pounds, and the demand is 10,600 pounds. When the price drops to $1.46 per pound, the supply decreases to 10,000...
-
Derive the equations for the stress-strain relationship, the creep compliance, and the relaxation modulus for the Zener model.
-
Ike issues $ 180,000 of 11%, three- year bonds dated January 1, 2013, that pay interest semiannually on June 30 and December 31. They are issued at $ 184,566. Their market rate is 10% at the issue...
-
You are a Morgan Stanley portfolio manager of a risky portfolio with an expected rate of return of 19% and a standard deviation of 28%. The T-bill rate is 7%. Suppose your client decides to invest in...
-
Twin-Cities, Inc., purchased a building for $600,000. Straight-line depreciation was used for each of the first two years using the following assumptions: 25-year estimated useful life, with a...
-
Suppose that the mass of the proton and the mass of the neutron were each exactly 1.0 u. What would the mass defect for a copper-63 nucleus be under these circumstances if its atomic mass is 62.5 u?
-
If the binding energy per nucleon (see the graph in Figure 11.16 ) increased steadily with ass number instead of peaking around A = 56, would nuclear fission and nuclear fusion reactions work the...
-
Your investor relations department reports to you that stockholders and financial analysts evaluate the quality of a company's financial reports based on their "transparency" namely the clarity and...
-
How do you interpret DD&A per BOE?
-
Which special costs or reserves must or may be included in amortization for full cost? Which costs or reserves must or may be included for successful efforts?
-
A company indicates in its 10K that it converted oil and gas to a common unit of measure by dividing its natural gas reserves and production by a factor of 5.81. That company is using the...
-
How do you interpret finding cost ratios? Why are they so popular in financial statement analysis?
-
When oil and gas are produced jointly, what basis or unit of measure is allowed for full cost amortization? When oil and gas are produced jointly, what basis or unit of measure is allowed for...
-
Provide an example in which the communication technology did not greatly affect the propinquity between you and another communicator. Now give an example in which it did affect the propinquity...
-
What does non-recourse financing mean?
-
The cross-section of a glass prism is an equilateral triangle. The refractive index of the glass is 1.601 at a free-space wavelength of 0.4 m and 1.569 at 0.7 m. Show that if white light is incident...
-
The following transfer function is not written in a standard form: (a) Put it in standard gain/time constant form. (b) Determine the gain, poles and zeros. (c) If the time-delay term is replaced by a...
-
For a leadlag unit, show that for a step input of magnitude M: (a) The value of y at t = 0 + is given by y(0+) = KM Ï a /Ï 1 . (b) Overshoot occurs only for Ï a > Ï 1 . (c)...
-
What effect does an antagonist drug have over the receptors ?
-
Read this article on the differences and Comparisons between UNIX and LINUX http://www.diffen.com/difference/Linux_vs_Unix, then do some research and post your thoughts on each of the systems. Based...
-
Assume that in 2023, Bobby had a vacant lot that was given to him as a gift from his father that had an FMV of $40,000 and an adjusted basis of $15,000.

Study smarter with the SolutionInn App


