A system formed initially of 2 mol CO 2 , 5 mol H 2 , and 1
Question:
A system formed initially of 2 mol CO2, 5 mol H2, and 1 mol CO undergoes the reactions:
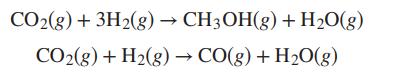
Develop expressions for the mole fractions of the reacting species as functions of the reaction coordinates for the two reactions.
Transcribed Image Text:
CO₂(g) + 3H2(g) → CH3OH(g) + H₂O(g) CO₂(g) + H₂(g) → CO(g) + H₂O(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To develop expressions for the mole fractions of the reacting species as functions of the reaction c...View the full answer

Answered By

User l_998468
I have extensive tutoring experience, having worked as a private tutor for over three years. I have tutored students from different academic levels, including high school, undergraduate, and graduate levels. My tutoring experience has taught me to be patient, attentive to student needs, and effective in communicating difficult concepts in simple terms.
I have a strong background in statistics, probability theory, data analysis, and data visualization. I am proficient in using statistical software such as R, Python, and SPSS, which are commonly used in academic research and data analysis. Additionally, I have excellent communication and interpersonal skills, which enable me to establish rapport with students, understand their learning styles, and adapt my teaching approach to meet their needs.
I am passionate about teaching and helping students achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781260597684
9th International Edition
Authors: J.M. Smith, Mark Swihart Hendrick C. Van Ness, Michael Abbott
Question Posted:
Students also viewed these Engineering questions
-
Develop expressions for the mole fractions of reacting species as functions of the reaction coordinate for: (a) A system initially containing 2 mol NH 3 and 5 mol O 2 and undergoing the reaction: 4NH...
-
A system initially containing 2 mol C 2 H 4 and 3 mol O 2 undergoes the reactions: C 2 H 4 (g)+O 2 (g)(CH 2 ) 2 O(g) C 2 H 4 (g)+3O 2 (g) 2CO 2 (g) +2H 2 O(g) Develop expressions for the mole...
-
Which of the following would be least important in the pursuit of a time-based strategy? A) flexible technology B) operational agility C) reduced complaint resolution times D) quick changeover times...
-
The correlation between the Charlottesville International Fund and the EAFE Market Index is 1.0. The expected return on the EAFE Index is 11%, the expected return on Charlottesville International...
-
The following data describe U.S. manufacturers shipments of general aviation jet aircraft from 1989 through 2008. a. Construct a graph of the time series. Does the overall trend appear to be upward...
-
Analyze the patient satisfaction data in Table B. 17 for multicollinearity. Satisfaction Age Severity Surgical-Medical Anxiety 68 55 50 0 2.1 77 46 24 1 2.8 96 30 46 1 3.3 80 35 48 1 4.5 43 59 58 0 2...
-
Presented here are an incomplete income statement and balance sheet for Schwenke Corporation. Additional information: 1. The gross profit margin is 40%. 2. The income tax rate is 20%. 3. The...
-
P3.1 (LO 1, 2, 3) (Multiple-Step Statement, Retained Earnings Statement) The following information is related to Dickinson Company for 2025. Retained earnings balance, January 1, 2025 Sales revenue...
-
Estelle owns a large concreting business as a sole trader. The business operates ten concreting trucks, each worth approximately $200,000, five of which Estelle holds freehold. Estelle would like to...
-
For the ammonia synthesis reaction written: With 0.5 mol N 2 and 1.5 mol H 2 as the initial amounts of reactants and with the assumption that the equilibrium mixture is in the ideal gas state, show...
-
Helium-laced gases are used as breathing media for deep-sea divers. Why? Table 13.2 may provide useful data. Table 13.2: Henry's Constants for Gases Dissolved in Water at 25C Gas Acetylene Air Carbon...
-
A binomial experiment consists of 500 trials. The probability of success for each trial is .4. What is the probability of obtaining the number of successes indicated in Problem? Approximate these...
-
State the objectives of government audit.
-
What are the steps to be taken by a statutory auditor before commencement of an audit of a company?
-
What do you understand by divisible profit? State what considerations should be borne in mind before declaring dividend.
-
State the matters required by the Companies Act, 1956 to be stated in auditors report to the shareholders on the accounts of a company audited by such auditor.
-
State the procedures an auditor should follow to verify the issue of share capital (a) For cash, (b) For consideration other than cash, and (c) For employees as sweat equity share.
-
A production line has four workstations and a 50- second cycle time. The total amount of actual task time across all four workstations is 170 seconds. What is the idle time? The percent idle time?...
-
TRUE-FALSE QUESTIONS 1. In terms of preliminary analytical procedures, assume that the company has introduced a new product with a low price point and significant customer demand. The auditor would...
-
Derive the form of the excess enthalpy predicted by Wilsons equation assuming that A ij s and ratios of molar volumes are temperature-independent.
-
Orbey and Sandler (1995. Ind. Eng. Chem. Res. 34:4351.) have proposed a correction term to be added to the excess Gibbs energy of mixing given by UNIQUAC. To a reasonable degree of accuracy the new...
-
Flash separations are fundamental to any process separation train. A full steady-state process simulation consists largely of many consecutive flash calculations. Use UNIFAC to determine the...
-
Assume that the City of Coyote has produced its financial statements for December 31, 2024, and the year then ended. The city's general fund was only used to monitor education and parks. Its capital...
-
Explain the difference between the mean, the median, and the mode.
-
Becky Beckles supports her Cousin Elle who lived with her all of 2018. Also three other persons but they donot live with he Joan Jones, who is Becky's mot Mom's husband anddaughter Laur Cousin Bill...

Study smarter with the SolutionInn App


