An ideal gas with constant heat capacities enters a converging/diverging nozzle with negligible velocity. If it expands
Question:
An ideal gas with constant heat capacities enters a converging/diverging nozzle with negligible velocity. If it expands isentropically within the nozzle, show that the throat velocity is given by: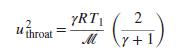 where T1 is the temperature of the gas entering the nozzle, ℳ is the molar mass, and R is the molar gas constant.
where T1 is the temperature of the gas entering the nozzle, ℳ is the molar mass, and R is the molar gas constant.
Transcribed Image Text:
yRT| throat r+1, (4)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
The velocity of a convergingdiverging nozzle can be found using the isentropic relation which states ...View the full answer

Answered By

Dulal Roy
As a tutor, I have gained extensive hands-on experience working with students one-on-one and in small group settings. I have developed the ability to effectively assess my students' strengths and weaknesses, and to customize my teaching approach to meet their individual needs.
I am proficient at breaking down complex concepts into simpler, more digestible pieces, and at using a variety of teaching methods (such as visual aids, examples, and interactive exercises) to engage my students and help them understand and retain the material.
I have also gained a lot of experience in providing feedback and guidance to my students, helping them to develop their problem-solving skills and to become more independent learners. Overall, my hands-on experience as a tutor has given me a deep understanding of how to effectively support and encourage students in their learning journey.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
An ideal gas with constant heat capacities undergoes a change of state from conditions T1, P1 to conditions T2, P2. Determine (H (J mol-1) and (S (J mol-1 K-1) for one of the following cases. (a) T1...
-
A mapping T: R2 R2 is given by Where Show that T rotates every vector v ε R2 counterclockwise about the origin through angle 0. Express v using polar coordinates, And use the identities...
-
An ideal gas with the adiabatic exponent goes through a cycle (Fig. 2.3) within which the absolute temperature varies -fold. Find the efficiency of this cycle.
-
Write a program to Sort the list U,N,I,V,E,R,S,I,T,Y in alphabetical order by Bubble sort I want the complete solution steps with the solution to the question
-
Name four of the positive participation skills required of team members.
-
The standard direct labor cost per reservation for Harrys Hotel is $3 (= $12 per labor hour ÷ 4 reservations per hour). Actual direct labor costs during the period totaled $45,240....
-
Compare and contrast prototyping and the incremental and iterative approach to software development.
-
Leisure is an inferior good for Horace. a. Use indifference curves to show the income and substitution effects of an increase in Horaces wage rate. b. Could Horaces labor supply curve be...
-
How do conflict resolution professionals navigate ethical dilemmas when mediating disputes between stakeholders with competing interests, balancing impartiality with the pursuit of equitable outcomes?
-
1. What are the factors driving the diversification of Tune Group? 2. What is the Tune Group portfolio, what contribution does each SBU make and, therefore, what is the dominant logic for this Group?...
-
A gas enters a converging nozzle at pressure P 1 with negligible velocity, expands isentropically in the nozzle, and discharges into a chamber at pressure P 2 . Sketch graphs showing the velocity at...
-
As suggested by Fig. 3.1, the slope of the sublimation curve at the triple point is generally greater than that of the vaporization curve at the same state. Rationalize this observation. Note that...
-
Otis Olson, the owner of two adjoining city lots, A and B, built a house on each. He laid a drainpipe from lot B across lot A to the main sewer pipe under the alley beyond lot A. Olson then sold and...
-
Write a stack client Parentheses that reads a string of parentheses, square brackets, and curly braces from standard input and uses a stack to determine whether they are properly balanced. For...
-
How much time does it take to call functions such as Math. \(\sin ()\), Math. \(\log ()\), and Math.sqrt()?
-
Write a filter Reverse that reads strings one at a time from standard input and prints them to standard output in reverse order. Use either a stack or a queue.
-
Why does allocating an array of length \(n\) take time proportional to \(n\) ?
-
Write a static method that reads floating-point numbers one at a time from standard input and returns an array containing them, in the same order they appear on standard input. Hint: Use either a...
-
The space R is defined as the set of all infinite real sequences a = (a1, a2, a3,...) where ai R. Define addition and scalar multiplication in such a way as to make R into a vector space. Explain...
-
Review Exhibit 11.4. Analyze each product on the graph according to the characteristics that influence the rate of adoption. For example, what can you conclude from the data about the relative...
-
Liquid/vapor saturation pressure P sat is often represented as a function of temperature by the Antoine equation, which can be written in the form: Here, parameters a, b, and c are substance-specific...
-
Electric current is the fundamental SI electrical dimension, with the ampere (A) as its unit. Determine units for the following quantities as combinations of fundamental SI units. (a) Electric power...
-
An incompressible fluid ( = constant) is contained in an insulated cylinder fitted with a frictionless piston. Can energy as work be transferred to the fluid? What is the change in internal energy of...
-
The weekly salaries (in dollars) of 24 randomly selected employees of a company are shown below: 310 320 450 460 470 500 520 580 600 650 700 540 710 840 870 900 1000 1200 1250 1300 1400 1720 2500...
-
(a) Assume a non-dividend paying asset St, undergoes geometric Brownian motion dS = S+dt+S+dWt where W is a standard Brownian motion and and are constants. We assume also that r, the risk-free...
-
Eleanor is auditing a large manufacturing company. Currently, she is looking at the entire population of inventory purchases for the year, and matching the purchase order, receiving documentation,...

Study smarter with the SolutionInn App


