For the system ethylene(1)/propylene(2) as a gas, estimate (a) Through application of Eqs. (10.63). (b) Assuming that
Question:
For the system ethylene(1)/propylene(2) as a gas, estimate
(a) Through application of Eqs. (10.63).
(b) Assuming that the mixture is an ideal solution.
![]()
(a) Eqs. (10.63)
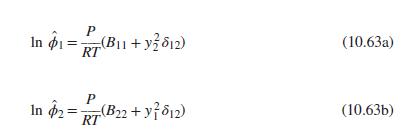
Transcribed Image Text:
fi. f2, ô1, and 2 at t = 150°C, P = 30 bar, and y1 = 0.35: %3D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
Equations 1063 are mu1 mu10 RTln a1 Delta1textex mu2 mu20 RTln a2 Delta2textex where mu1 and mu2 ar...View the full answer

Answered By

FELIX NYAMBWOGI
I have been tutoring for over 5 years, both in person and online. I have experience tutoring a wide range of subjects, including math, science, English, and history. I have also worked with students of all ages, from elementary school to high school.
In addition, I have received training in effective tutoring strategies and techniques, such as active listening, questioning, and feedback. I am also proficient in using online tutoring platforms, such as Zoom and Google Classroom, to effectively deliver virtual lessons.
Overall, my hands-on experience and proficiency as a tutor has allowed me to effectively support and guide students in achieving their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
An ideal gas refrigeration system with two stages of compression with intercooling as shown in Fig. P11-77 operates with air entering the first compressor at 90 kPa and - 24oC. Each compression stage...
-
An ideal gas is taken through a Carnot cycle. The isothermal expansion occurs at 250C, and the isothermal compression takes place at 50.0C. The gas takes in 1 200 J of energy from the hot reservoir...
-
An ideal gas is taken Figure from a to b on the pV-diagram shown in Fig. 19.22. During This process, 400 J of heat is added and the pressure doubles. (a) How much work is done by or on the gas?...
-
The unadjusted trial balance of Mesa Inc., at the company's year end of December 31 follows: Additional information and adjustment data: 1. The 12-month insurance policy was purchased and was...
-
Use the following information to answer (a) (c): (a) Between Thursday and Friday, did the U.S. dollar appreciate or depreciate against the Russian ruble? (b) Between Thursday and Friday, did the U.S....
-
The supplies account had a beginning balance of $2,400 and was debited for $3,975 for supplies purchased during the year. Journalize the adjusting entry required at the end of the year assuming the...
-
What are the main differences between sequence and collaboration diagrams? When would you use each?
-
David R. and Ella M. Cole (ages 39 and 38, respectively) are husband and wife who live at 1820 Elk Avenue, Denver, CO 80202. David is a regional sales manager for Wren Industries, a national...
-
Required information Problem 6-2AA (Algo) Periodic: Alternative cost flows LO P3 [The following information applies to the questions displayed below] Warnerwoods Company uses a periodic inventory...
-
A plant manager is considering investing in a new $30,000 machine. Use of the new machine is expected to generate a cash flow of about $8,000 per year for each of the next five years. However, the...
-
Make use of Eqs. (3.36), (3.61), (3.62), (6.54), (6.55), (6.56), (6.70), (6.71), (10.62), and (10.69)(10.74), to estimate V, HR, SR, and GR for one of the following binary vapor mixtures: (a)...
-
Justify the following equations: ( - VR aln T P.x RT RT2 T.x GR Ex, In Exd In = 0 (const T, P) RT
-
If reserves increase by $2 million and the required reserve ratio is 8 percent, what is the change in the money supply?
-
Navigation from one page to another can be done in two ways. One is directly to use code by writing a JSP tag such as or an HTML hyperlink in the JSF file. Another way that is provided by JSF is to...
-
UsePL-SQL to create a package, Student_Course, in the Oracle SQL Developer. The package contains a stored procedure namedSelectStudentCourse() with two positional parameters, a. The input student...
-
When using the JFileChooser object to select an image, the returned image is a Java File object, and one needs to convert it to a Byte[] array to be inserted into a column in a database Table....
-
To update a record using the UpdaTable ResultSet, one needs to use steps, and they are: . a. 1, UpdateXXX() b. 2, UpdateXXX() and UpdateRow() c. 3, UpdateXXX(), UpdateCursor() and UpdateRow() d. 4,...
-
a. Why does the federal government subsidize farmers? b. Explain how a subsidy paid to cotton farmers affects the price of cotton and the marginal cost of producing it. Congress sent the White House...
-
(a) Are there any nonzero real scalars that satisfy (a + b)-l = a-l + B-l? (b) Are there any nonsingular real 22 matrices that satisfy (A + B)-1 = A-1 + B-1?
-
By referring to Figure 13.18, determine the mass of each of the following salts required to form a saturated solution in 250 g of water at 30 oC: (a) KClO3, (b) Pb(NO3)2, (c) Ce2(SO4)3.
-
A turbine operates adiabatically with superheated steam entering at T1 and P1 with a mass flow rate m. The exhaust pressure is P2 and the turbine efficiency is . For OIK of the following sets of...
-
A turbine operates adiabatically with superheated steam entering at T1 and P1 with a mass flow rate m. The exhaust pressure is P2 and the turbine efficiency is . For OIK of the following sets of...
-
Nitrogen gas initially at 8.5 bar expands isentropically to 1 bar and 150 C Assuming nitrogen to be an ideal gas, calculate the initial temperature and the work produced per mole of nitrogen.
-
Recommend an optimal improvement to the company's future financial goals.
-
Your bank account pays a nominal interest rate of 1 1 % , compounded semi - annually. Your plan is to deposit $ 2 , 4 0 0 in the account at the end of each of the next 1 0 years. How much will you...
-
A partnership has total equity of $464,000. Partnership equity consists of Green, Capital, $324,000, and Tan, Capital, $140,000. Net income and loss is shared in a ratio of 85% to Green and 15% to...

Study smarter with the SolutionInn App


