For water at each of the following states, determine the internal energy and enthalpy using the steam
Question:
For water at each of the following states, determine the internal energy and enthalpy using the steam tables.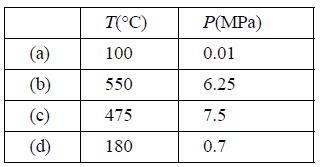
Transcribed Image Text:
(a) (b) (c) | 0 T(C) 100 550 475 180 P(MPa) 0.01 6.25 7.5 0.7
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
To determine the internal energy U and enthalpy H of water at the given states using steam table...View the full answer

Answered By

Deepak Pal
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students. Areas of interest: Business, accounting, Project management, sociology, technology, computers, English, linguistics, media, philosophy, political science, statistics, data science, Excel, psychology, art, history, health education, gender studies, cultural studies, ethics, religion. I am also decent with math(s) & Programming. If you have a project you think I can take on, please feel welcome to invite me, and I'm going to check it out!
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemical Engineering Thermodynamics
ISBN: 9780136068549
2nd Edition
Authors: J. Elliott, Carl Lira
Question Posted:
Students also viewed these Engineering questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Determine the change in internal energy and enthalpy of water at 100°F and 500 psia if the temperature increases to 500°F while the pressure in Fig. 2.34 is held constant. Use a) The steam...
-
Determine whether water at each of the following states is a compressed liquid, a superheated vapor, or a mixture of saturated liquid and vapor.
-
Track a futures contract for 5 consecutive days record the prices and write a short summary (150-200 words) describing how and why prices moved the way they did. You will be graded upon correctly...
-
If you are able to save $5000 in year 1, $5150 in year 2, and amounts increasing by 3% each year through year 20, the amount you will have at the end of year 20 at 10% per year interest is closest...
-
At what stage should you begin to think about the kinds of data analysis you need to conduct?
-
What is contribution?
-
The Lubbock plant of Morrils Small Motor Division produces a major subassembly for a 6.0 horsepower motor for lawn mowers. The plant uses a standard costing system for production costing and control....
-
Note :- First picture is of the question and other are for reference. Note:- I want full answer not just hints. 2. b) Based on Mr. Suzuki's estimate of a 3-5\% growth rate of Saito Solar's free cash...
-
(a) What size vessel holds 2 kg water at 80C such that 70% is vapor? What are the pressure and internal energy? (b) A 1.6 m 3 vessel holds 2 kg water at 0.2 MPa. What are the quality, temperature,...
-
A permeation membrane separates an inlet air stream, F, (79 mol% N 2 , 21 mol% O 2 ), into a permeate stream, M, and a reject stream, J. The inlet stream conditions are 293 K, 0.5 MPa, and 2 mol/min;...
-
Sodium ions, Na + , form ionic compounds with fluoride ions, F , and iodide ions, I . The radii of these ions are as follows: Na + = 116 pm; F = 119 pm; and I = 206 pm. In which ionic compound,...
-
# III: Worksheet 3 1. A 20 kg mass is allowed to accelerate down a frictionless 15 ramp. 20 kg 15 a. Draw a force diagram for the block. b. Determine the value of the x-component of the force of...
-
3.Baker Corporation has provided the following production and average cost data for two levels of monthly production volume. The company produces a single product Production Volume: 1,000 units:...
-
Suppose that you own the only company in the market to produce a certain product, and therefore you can determine the market price P dollars for each unit. Due to government regulations, the price of...
-
describes how the blast pressure front can bounce off solid, immovable obstacles and be redirected in another direction in a linear angle to the angle of the obstacle hat was struck
-
As the accounting clerk, you are tasked by the CFO to determine the cost of goods sold of Del Mundo Company for the year ended December 31, 2020. During Operating cost data annd inventory account...
-
ABC Inc. is in the business of airframe maintenance, modification and retrofit services, avionics and aircraft interior installations, the overhaul and repair of aircraft engines, and other related...
-
A city maintains a solid waste landfill that was 12 percent filled at the end of Year 1 and 26 percent filled at the end of Year 2. During those periods, the government estimated that total closure...
-
For a gas described by the Redlich/Kwong equation and for a temperature greater than T c , develop expressions for the two limiting slopes, Note that in the limit as P 0, V , and that in the limit...
-
For a gas described by the Redlich/Kwong equation and for a temperature greater than T c , develop expressions for the two limiting slopes, Note that in the limit as P 0, V , and that in the limit...
-
Figure 3.3 suggests that the isochores (paths of constant volume) are approximately straight lines on a P-T diagram. Show that the following models imply linear isochores. (a) Constant-, equation...
-
Ted and his partners have contracted to purchase the franchise nights worth 561 000 to open and operate a specialty pizza restaurant called Popper with a renewable agrement, the partners have agreed...
-
Your answer is partially correct. Martin Company's chief financial officer feels that it is important to have data for the entire quarter especially since their financial forecasts indicate some...
-
Kellog Corporation is considering a capital budgeting project that would have a useful life of 4 years and would love testing 5156.000 in equipment that would have zeto salvage value at the end of...

Study smarter with the SolutionInn App


