The pressure above a mixture of ethanol and ethyl acetate at 70C is measured to be 78
Question:
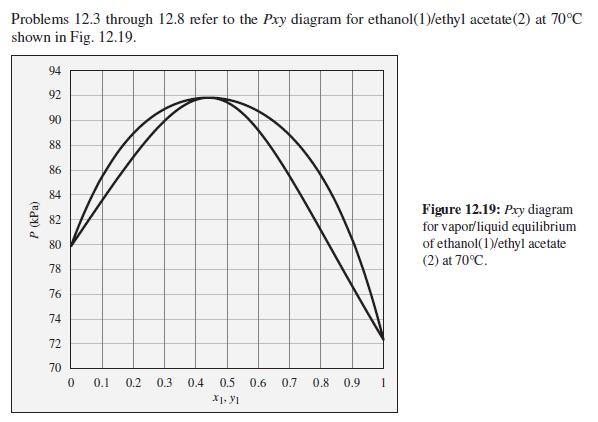
The pressure above a mixture of ethanol and ethyl acetate at 70°C is measured to be 78 kPa. What are the possible compositions of the liquid and vapor phases?
Transcribed Image Text:
Problems 12.3 through 12.8 refer to the Pxy diagram for ethanol(1)/ethyl acetate(2) at 70°C shown in Fig. 12.19. 94 92 90 88 86 84 Figure 12.19: Pry diagram for vapor/liquid equilibrium of ethanol(1)/ethyl acetate (2) at 70°C. 82 80 78 76 74 72 70 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 X1. y1 P (kPa)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
A relativistic rocket is measured to be 50 m long, 2.5 m high, and 2.0 m wide by its pilot. It is traveling at 0.65c (in the direction parallel to its length) relative to an inertial observer. (a)...
-
A mixture of ethanol and 1-propanol behaves ideally at 36C and is in equilibrium with its vapor. If the mole fraction of ethanol in the solution is 0.62, calculate its mole fraction in the vapor...
-
The pressure of an automobile tire is measured to be 190 kPa (gage) before a trip and 215 kPa (gage) after the trip at a location where the atmospheric pressure is 95 kPa. If the temperature of air...
-
Count Dracula, the most famous vampire, rumored to have killed at least 200,000 people, was based on a real person who lived in eastern Europe about 600 years ago. He was indeed a "monster," although...
-
Is it possible for everyones real income to rise even though the income distribution in a society has become more unequal? Prove your answer with a numerical example.
-
Balance sheet data for Kamel Corporation follow Current assets ............ $ 240,000 Long-term assets (net) ........... 760,000 Total assets ............. $1,000,000 Current liabilities ..............
-
The price of a share is \(\$ 40\). If \(\mu=0.1\) and \(\sigma^{2}=0.16\) per year, find a \(95 \%\) confidence interval for the price of the share after six months (i.e. an interval...
-
Following are the balance sheets for the years ended June 30, 2018 and 2017 for Seahorse Inc.: Required: a. Calculate the current ratio and the quick ratio on June 30, 2017 and 2018. b. Assess the...
-
Write code for the same thing exactly; send me the code so I can run it and send me the output. This is for engineers who understand networking and programming; if you are not one, kindly do not...
-
Brothers Harry and Herman Hausyerday began operations of their machine shop (H & H Tool, Inc.) on January 1, 2010. The annual reporting period ends December 31. The trial balance on January 1, 2012,...
-
Consider an ethanol(1)/ethyl acetate(2) mixture with x 1 = 0.70, initially at 70C and 100 kPa. Describe the evolution of phases and phase compositions as the pressure is gradually reduced to 70 kPa....
-
Of the following binary liquid/vapor systems, which can be approximately modeled by Raoults law? For those that cannot, why not? Table B.1 (App. B) may be useful. (a) Benzene/toluene at 1(atm). (b)...
-
Calculate the pH of a 1 L solution containing (a) 10 mL of 5 M NaOH, (b) 10 mL of 100 mM glycine and 20 mL of 5 M HCl, and (c) 10 mL of 2 M acetic acid and 5 g of sodium acetate (formula weight 82 g ...
-
On January 1, 1985, Ivan Innkeeper purchased an office building for $900,000. He used the ACRS statutory (accelerated) depreciation method and took $900,000 depreciation before he sold the building...
-
Irene Irwin gave her son James a Section 1245 machine. The machine has an adjusted basis of $25,000 and Irene had taken $10,000 in depreciation. James used the machine for 15 months and took $5,000...
-
Mary Martin owns a custom curtain/drapery business. In 2019, she purchased three new sewing machines and in a separate transaction, sold her three old machines for $6,000. She had bought the old...
-
Marielle Machinery Works is considering a project which has an initial investment of 10,000 and has expected cash flows of 0 in year 1, 7,500 in year 2, and 8,500 in year 3. The company uses the IRR...
-
Etonic SA is considering an investment of 250,000 in an asset with an economic life of 5 years. The firm estimates that the nominal annual cash revenues and expenses at the end of the first year will...
-
Find a unitary matrix p that diagonalizes A, and determine P-1 AP. 7. 9. 11. 4 1-i 1+i 5 T6 2+i 2-2i 4 3 A-0 11+i 01-i 0
-
How much more interest will be earned if $5000 is invested for 6 years at 7% compounded continuously, instead of at 7% compounded quarterly?
-
What is the composition of the vapor phase in equilibrium with a liquid phase chloroform(1)/tetrahydrofuran(2) mixture of the following compositions at P = 1 bar? (a) x 1 = 0.1 (b) x 1 = 0.2 (c) x 1...
-
What is the composition of the liquid phase in equilibrium with a vapor phase ethanol(1)/ethyl acetate(2) mixture of the following compositions at P = 1 bar? (a) y 1 = 0.1 (b) y 1 = 0.2 (c) y 1 = 0.3...
-
What is the composition of the vapor phase in equilibrium with a liquid phase ethanol(1)/ethyl acetate(2) mixture of the following compositions at P = 1 bar? (a) x 1 = 0.1 (b) x 1 = 0.2 (c) x 1 = 0.3...
-
KORBIN COMPANY Comparative Income Statements For Years Ended December 31 2021 2020 $ 316,588 2019 $ 219,700 Sales Cost of goods sold Gross profit Selling expenses Administrative expenses Total...
-
Following are the capital account balances and profit and loss percentages (indicated parenthetically) for the Alden, Boyd, and Carmel partnership: Alden (40%) Boyd (40%) Carmel (20%) $ 220,000...
-
Required information E3-13 (Algo) Recording and Posting Accrual Basis Journal Entries, and Preparing an Unadjusted Trial Balance and Preliminary Financial Statements [LO 3-2, 3-3, LO 3-4] [The...

Study smarter with the SolutionInn App


