Consider an ethanol(1)/ethyl acetate(2) mixture with x 1 = 0.70, initially at 70C and 100 kPa. Describe
Question:
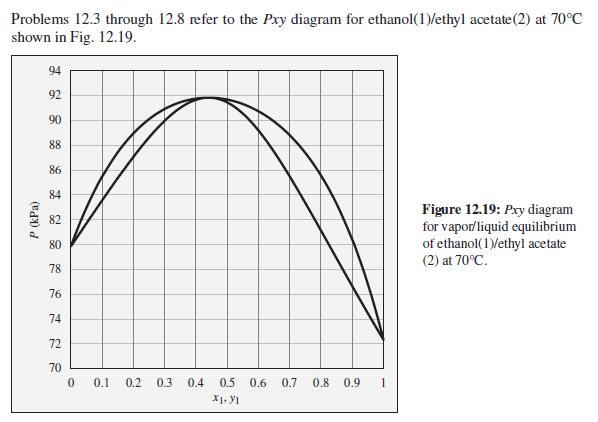
Consider an ethanol(1)/ethyl acetate(2) mixture with x1 = 0.70, initially at 70°C and 100 kPa. Describe the evolution of phases and phase compositions as the pressure is gradually reduced to 70 kPa.
Transcribed Image Text:
Problems 12.3 through 12.8 refer to the Pxy diagram for ethanol(1)/ethyl acetate(2) at 70°C shown in Fig. 12.19. 94 92 90 88 86 84 Figure 12.19: Pry diagram for vapor/liquid equilibrium of ethanol(1)/ethyl acetate (2) at 70°C. 82 80 78 76 74 72 70 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 X1. y1 P (kPa)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
To describe the evolution of phases and phase compositions as the pressure is gradually reduced we need to construct the Pxy diagram for the given eth...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
Describe the evolution of business-to-business (B2B) e-commerce.
-
Describe the evolution of ERP systems.
-
Describe the evolution of American culinary arts.
-
In 2014, Elbert Corporation had net cash provided by operating activities of $531,000; net cash used by investing activities of $963,000; and net cash provided by financing activities of $585,000. At...
-
1. Explain what economists mean when they refer to interest. 2. Listthe components of demand for loanable funds. 3. List the components of supply of loanable funds. 4. Graphically show the loanable...
-
Many service industries link labor rate and time (quantity) standards with their processes. One example is the standard time to board an aircraft. The reason time plays such an important role in the...
-
Suppose that the price of a share verifies that \(\mu=16 \%\) and the volatility is \(30 \%\). If the closing price of the share at a given day is \(\$ 50\), compute: (a) The closing expected value...
-
For each of the following specific audit procedures, indicate the type of audit procedure it represents: (1) Inspection of records or documents, (2) Inspection of tangible assets, (3) Observation,...
-
Type cd and then hit enter key. Now, create a directory lab2 and perform following steps in lab2 directory. root bin etc users tmp usr john work work carol play 1. Create the directory tree depicted...
-
Use the following information to prepare a 2021 Form 1040, Schedule 1, and Schedules A and B for Leonard Cox. 1. Leonard Cox is a single taxpayer. Leonards social security number is 445-01- 9266, and...
-
What is the composition of the azeotrope for the ethanol(1)/ethyl acetate(2) system? Would this be called a high-boiling or low-boiling azeotrope? Problems 12.3 through 12.8 refer to the Pxy diagram...
-
The pressure above a mixture of ethanol and ethyl acetate at 70C is measured to be 78 kPa. What are the possible compositions of the liquid and vapor phases? Problems 12.3 through 12.8 refer to the...
-
An investment bank pays $ 23.50 per share for 3,000,000 shares of the KDO Company. It then sells these shares to the public for $ 25. How much money does KDO receive? What is the investment bankers...
-
Peter Paterson transfers an apartment building with an adjusted basis of $160,000 and a fair market value of $235,000 for Charlie Claussen's apartment building (adjusted basis $120,000) with a fair...
-
Carl Cushman, a college professor, age 58, purchased and moved into a house on August 1, 2017. He used the house continuously until September 1, 2018, on which date he went abroad for a one-year...
-
Jim Junction purchased a building business on November 17, 2018, for $400,000. On July 21, 2019, he exchanged the building for another business building in a like-kind exchange. The new building had...
-
Gordon Grumps is married and files separately. During 2019, he had the following capital gains and losses: Gordons taxable income is $6,000. What is Gordon's capital loss deduction for 2019 and his...
-
Alice Almond purchased Smith Corporation stock on February 23, 2018, and on November 15, 2018, she gave the stock to her son Dennis. She paid $10,000 for the stock and the value at the time of the...
-
Show that the eigenvalues of a unitary matrix have modulus 1.
-
A statistical study shows that the fraction of television sets of a certain brand that are still in service after x years is given by f (x) = e-0.15x. (a) What fraction of the sets are still in...
-
The following liquids, all at atmospheric pressure and 300 K, are mixed: 25 kg of pure water, 40 kg of pure sulfuric acid, and 75 kg of 25-wt-% sulfuric acid. (a) How much heat is liberated if mixing...
-
Ten thousand (10,000) kgh 1 of an 80-wt-% H 2 SO 4 solution in water at 300 K is continuously diluted with chilled water at 280 K to yield a stream containing 50-wt-% H 2 SO 4 at 330 K. (a) What is...
-
What is the composition of the liquid phase in equilibrium with a vapor phase chloroform(1)/tetrahydrofuran(2) mixture of the following compositions at P = 1 bar? (a) y 1 = 0.1 (b) y 1 = 0.2 (c) y 1...
-
E3-20 (Algo) Analyzing, Recording, and Evaluating the Effects of Income Statement Transactions [LO 3-3, LO 3-4, LO 3-5] [The following information applies to the questions displayed below.] MyBnB...
-
Required information [The following information applies to the questions displayed below.] The following data reports on the July production activities of the Molding department at Ash Company....
-
Question 1 Vieux Enterprise's total overhead costs at various levels of activity follow: Month Machine- Hours Total Overhead Costs April 70,000 $198,000 May 100,000 270,000 June 80,000 222,000 July...

Study smarter with the SolutionInn App


