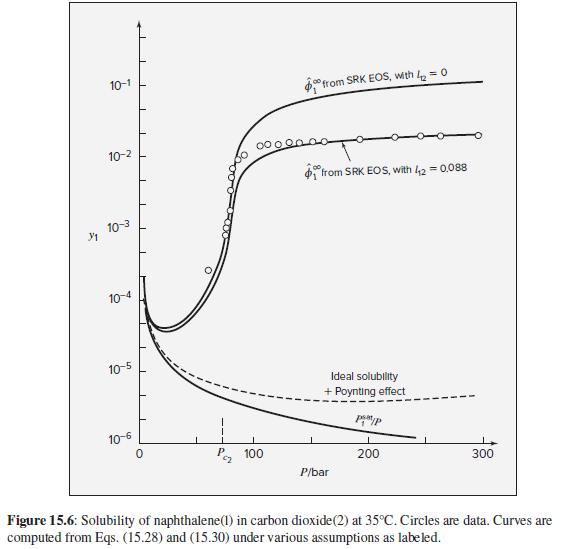
The qualitative features of SVE at high pressures shown in Fig. 15.6 are determined by the equation
Question:
The qualitative features of SVE at high pressures shown in Fig. 15.6 are determined by the equation of state for the gas. To what extent can these features be represented by the two-term virial equation in pressure, Eq. (3.36)?
Figure 15.6
Eq (3.36)

Transcribed Image Text:
10-1 °from SRK EOS, with k = 0 10-2 from SRK EOS, with 42 = 0.088 10-3 10-4 10-5 Ideal solubility + Poynting effect 10-6 P, 100 200 300 P/bar Figure 15.6: Solubility of naphthalene(1) in carbon dioxide(2) at 35°C. Circles are data. Curves are computed from Eqs. (15.28) and (15.30) under various assumptions as labeled.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (14 reviews)
The question is regarding the solubility of naphthalene in carbon dioxide at high pressures and how well this can be described by a twoterm virial equ...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
What are a firm's dynamic capabilities? To what extent can managers create or "manage into existence" a firm's dynamic capabilities?
-
To what extent can personal creditors seek recovery from partnership assets?
-
To what extent can government regulate individual choices regarding sexuality?
-
A ball, which we can treat as a point charge, has a charge of +Q. This ball is 50 cm away from a ball of charge-100, which is fixed in position. The +Q ball is 30 cm vertically below, and 40 cm...
-
Excess capacity is the price we pay for production differentiation. Evaluate this statement in terms of monopolistic competition.
-
How do fundamental analysis and technical analysis differ?
-
Based on information in this chapter, recommend the most effective HR administrative approach or approaches for the owner of a small business with fewer than 50 employees and infrequent staffing...
-
Harrison Electronics, Inc. operates a chain of electrical lighting and fixture distribution centers throughout northern Arizona. The firm is anticipating expansion of its sales in the coming year as...
-
Sit Down Publishers publishes The Accountant magazine and sells one - year ddsubscriptions to its customers. Customers are required to pay in full for the one - year subscription at the beginning of...
-
The head of a citys Recreation Department feels that the employees of the department are the best in the city. Each year, all city employees receive a standardized performance evaluation that rates...
-
Any equation of state valid for gases in the zero-pressure limit implies a full set of virial coefficients. Show that the second and third virial coefficients implied by the generic cubic equation of...
-
Work the preceding problem for mole fractions z 1 = 0.32, z 2 = 0.45, z 3 = 0.23.
-
Find f(x). f(x) = 1/(5x 3) 4 and f(1) = 90.
-
Each instance that follows is an example of one of the four types of market failure discussed in this chapter. In each case, identify the type of market failure and defend your choice briefly. a....
-
New PhDs in economics entering the job market find that academic jobs (jobs teaching at colleges and universities) pay about 30 percent less than nonacademic jobs such as working at a bank or a...
-
Which of the following are actual Pareto-efficient changes? Explain briefly. a. You buy an air conditioner as it is hotter this summer due to climate change. b. You go salmon-fishing but you throw...
-
According to the United Airlines Website, their Basic Economy fare differs from their standard Economy fare as follows: complimentary seat selection and upgrades are not available, group and family...
-
In a significant decision regarding patent eligibility, Alice Corp. v. CLS Bank International, the U.S Supreme Court in June 2004 effectively raised the bar for software patents by declining a patent...
-
Show that every solution of y = ay has the form y = ceax.
-
Graph the following conic sections, labeling vertices, foci, directrices, and asymptotes (if they exist). Give the eccentricity of the curve. Use a graphing utility to check your work. 10 5 + 2 cos 0
-
Laugier and Richon (J. Chem. Eng. Data, 40:153, 1995) report the following data for the H 2 S + benzene system at 323 K and 2.010 MPa: x 1 = 0.626; y 1 = 0.986. (a) Quickly estimate the vapor-liquid...
-
Consider two gases that follow the virial equation. Show that an ideal mixture of the two gases follows the relation B = y 1 B 11 + y 2 B 22 .
-
Acrolein (C 3 H 4 O) + water exhibits an atmospheric (1 bar) azeotrope at 97.4 wt% acrolein and 52.4C. For acrolein: T c = 506 K; P c =51.6 bar; and = 0.330; MW =56. (a) Determine the value of k ij...
-
Example: Make vs Buy: A decision concerning whether an item should be produced internally or purchased from an outside supplier. Java J's is a coffee shop offering a variety of coffees, teas and...
-
3. An elastic material fills a cubic-shaped cavity in a rigid metal form. The properties of the material are: E = 15000 MPa v = 0.25 = 120 10-6/C (coefficient of thermal expansion) The material is...
-
Every decision has an Opportunity Cost due to the nature of scarcity, there is always a better alternative not chosen, therefore, there is always an opportunity cost. "The opportunity cost of an...

Study smarter with the SolutionInn App


