(a) Use the ClausiusClapeyron equation and data for water at 100C to develop an expression for the...
Question:
(a) Use the Clausius–Clapeyron equation and data for water at 100°C to develop an expression for the vapor pressure of water as a function of temperature.
(b) Plot the expression you came up with on a PT diagram for temperatures from 0.01°C to 100°C.
(c) Include data from the steam tables on your plot in part
(b) and comment on the adequacy of the Clausius–Clapeyron equation.
(d) Repeat parts (b) and (c) for 100°C to 200°C.
(e) Repeat parts (a)–(c), but correct for the temperature dependence of Dhvap, using heat capacity data from Appendix A.2.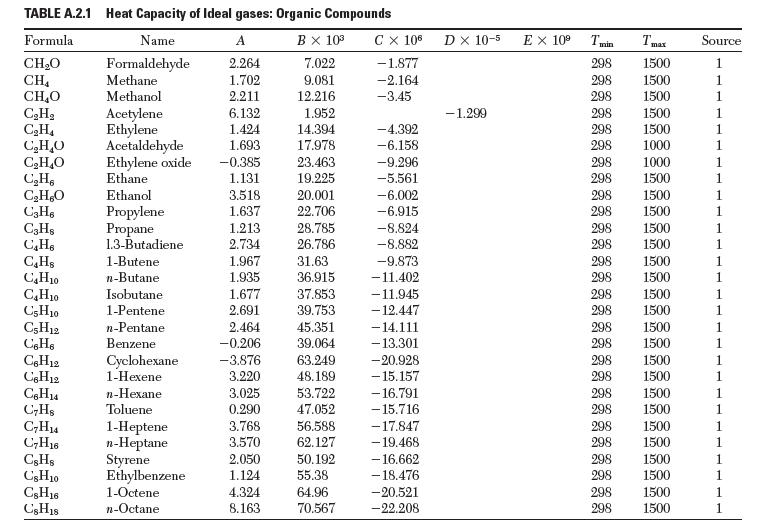
Transcribed Image Text:
TABLE A.2.1 Heat Capacity of Ideal gases: Organic Compounds Formula Name B X 10 CHO 7.022 Formaldehyde Methane CH 9.081 CHO Methanol 12.216 CH CH4 CHO CHO CH6 CHO C3H6 C3Hs C4H C4Hs C4H10 CH0 C3H10 C5H12 C6H6 C6H12 C6H12 C6H4 C7Hs C7H4 C7H16 CsHs CsH10 CsH16 CsH18 Acetylene Ethylene Acetaldehyde Ethylene oxide Ethane Ethanol Propylene Propane 1.3-Butadiene 1-Butene n-Butane Isobutane 1-Pentene n-Pentane Benzene Cyclohexane 1-Hexene n-Hexane Toluene 1-Heptene n-Heptane Styrene Ethylbenzene 1-Octene n-Octane A 2.264 1.702 2.211 6.132 1.424 1.693 -0.385 1.131 3.518 1.637 1.213 2.734 1.967 1.935 1.677 2.691 2.464 -0.206 -3.876 3.220 3.025 0.290 3.768 3.570 2.050 1.124 1.952 14.394 17.978 23.463 19.225 20.001 22.706 28.785 26.786 31.63 36.915 37.853 39.753 45.351 39.064 63.249 48.189 53.722 47.052 56.588 62.127 50.192 55.38 4.324 64.96 8.163 70.567 C X 106 -1.877 -2.164 -3.45 -4.392 -6.158 -9.296 -5.561 -6.002 -6.915 -8.824 -8.882 -9.873 -11.402 -11.945 -12.447 - 14.111 -13.301 -20.928 -15.157 -16.791 -15.716 -17.847 - 19.468 - 16.662 - 18.476 -20.521 -22.208 D X 10-5 EX 10 -1.299 298 298 298 298 298 298 298 298 298 298 298 1500 298 1500 298 1500 298 1500 1500 1500 298 298 298 298 298 298 298 298 298 298 Tmax 1500 1500 1500 298 298 298 298 1500 1500 1000 1000 1500 1500 1500 1500 1500 1500 1500 1500 1500 1500 1500 1500 1500 1500 1500 Source 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Processing times (including setup times) for five jobs that arrived today and are waiting to be processed in the same order on three machines in a shop are given below. Determine the sequence of jobs...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Portray in words what transforms you would have to make to your execution to some degree (a) to accomplish this and remark on the benefits and detriments of this thought.You are approached to compose...
-
Certain companies regard the management of cultural diversity as a way of developing new competencies that give them certain advantages. They are, for example, able to recruit and hold on to good...
-
Identify a company-either a publicly-traded company whose recent newsworthy stories have gained your attention or your own organization -that is in need of advice for how their HR professionals might...
-
Write the expression in the standard form a + bi. 15 21 cos 10 m + i sin 10
-
Two identical fans were tested for motor inrush current. One motor would trip after taking too long to achieve full speed, whereas the other motor exhibited no issues. See below: Current inrush on...
-
Who is the principal, and who is the agent in this scenario? By which method was an agency relationship formed between Scott and Blatt? James Blatt hired Marilyn Scott to sell insurance for the...
-
What are the thermodynamic and kinetic considerations in ultrasonic-assisted extraction, and how does this method enhance the efficiency and speed of extraction compared to conventional techniques ?
-
What pressure is needed to isothermally compress ice initially at 25C and 1 bar so that it changes phase?
-
For a given binary system, the partial molar volume of species 1 is constant. What can you say about species 2? Explain.
-
For-profit organizations face a requirement to earn at least a minimum-level return on investment. Some businesses rely on high ratios of income to sales; other businesses rely on high ratios of...
-
Mike is doing some holiday shopping. In most stores, he uses his bank card to pay. In some stores, he uses cash. Mike withdraws $80 cash from his account. The ATM receipt says his balance is $57.30....
-
How are events in Eastern Europe of East Asia (Ukraine and Taiwan) affecting financial markets? What financial market indicators reflect the seriousness? (answer to be at least a paragraph)
-
Find on the curve x^2/4-8y^2-25 the closest points to the point (0,0). Find the total derivative df/dt: (a) f(x,y)= t^2 (y-4x)^3, where x(t)=ln(6t), and y(t)=4e^(t/2) (b) f(x,y)=ln(xy^2)+e^(x/y)-t,...
-
Community General Hospital has enjoyed a history of success in attracting and maintaining solid patient volumes. Part of their success has been a balanced program of capital equipment purchases to...
-
The internal rate of return on a property purchased for $100,000 cash and generating $10.000 net annual cash flows for 10 years with a $ 100,000 net cash reversion expected at the end of the lease...
-
Ethical Issue: Should employers have the right to monitor all e-mail and instant messages that employees exchange on the job? Present the employers position and then the employees position.
-
CRUZ, INC. Comparative Balance Sheets December 31, 2015 CRUZ, INC. Income Statement For Year Ended December 31, 2015 Required Use the indirect method to prepare the cash provided or used from...
-
The No molecule has a doubly degenerate excited electronic level 121.1 cm-1 above the doubly degenerate electronic ground term. Calculate and plot the electronic partition function of NO from T= 0 to...
-
Calculate, by explicit summation, the vibrational partition function and the vibrational contribution to the molar internal energy of is molecules at (a) 100 K, (b) 298 K given that its vibrational...
-
A sample consisting of five molecules has a total energy SE. Each molecule is able to occupy states of energy jE, with j = 0, 1,2, .... (a) Calculate the weight of the configuration in which the...
-
1. Why is it important to review existing policies before shopping for new coverage? 2. What are the advantages and disadvantages of different types of insurance intermediaries from the perspective...
-
What role do insurance companies play in the financial education of Honduran citizens? Should they take a more active role in this sense?
-
3. (20p) Assume that you manage a dry cleaning business. Using the regression analysis tool in Excel to estimate your firm's demand curve (for cleaning 2-piece suits), you obtained the following...

Study smarter with the SolutionInn App


