The following data are available for vaporliquid equilibrium of the methane (1) n-pentane (2) binary system at
Question:
The following data are available for vapor–liquid equilibrium of the methane (1) –n-pentane (2) binary system at 37.78°C. Compare how well the van der Waals and Peng–Robinson equations can represent these data using the equation of state method to calculate fugacity coeffi cients of the vapor and the liquid.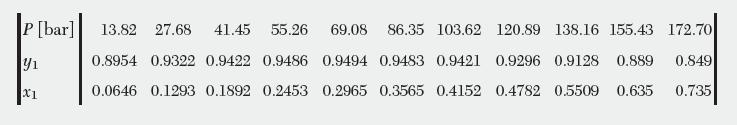
Transcribed Image Text:
P [bar] 13.82 27.68 41.45 55.26 69.08 86.35 103.62 120.89 138.16 155.43 172.70 0.849 0.8954 0.9322 0.9422 0.9486 0.9494 0.9483 0.9421 0.9296 0.9128 0.889 0.0646 0.1293 0.1892 0.2453 0.2965 0.3565 0.4152 0.4782 0.5509 0.635 0.735 y X1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
We can use the solution approach developed in Examples 815 and 816 to calculate the value of pressur...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Repeat Example 8.17 for the PengRobinson equation with values of the binary interaction parameter, k12 of 0.025, 0.05, and 0.10. Example 8.17 The following data are available for vaporliquid...
-
Compute the CLV metric and then apply it in a simple breakeven analysis. The data below show the results of a Starbucks segmentation study. Each column shows the values for a typical customer in each...
-
The directors of Mylo Ltd are currently considering two mutually exclusive investment projects. Both projects are concerned with the purchase of new plant. The following data are available for each...
-
Prepare adjusting journal entries, as needed, considering the account balances excerpted from the unadjusted trial balance and the adjustment data. A. depreciation on fixed assets, $ 8,500 B....
-
Which of the following questions should a marketing researcher ask when trying to establish the reliability of secondary data sources in the international arena? a. How much does the data cost? b....
-
Suppose you use a second-order Taylor polynomial centered at 0 to approximate a function f . What matching conditions are satisfied by the polynomial?
-
Sample office rental rates (in dollars per square foot per year) for Los Angeles are shown in the table at the left. Use technology to find the mean rental rate and the sample standard deviation.
-
Crane Corporation has the following accounts included in its December 31, 2012, trial balance: Equity Investments (trading) $21,000; Goodwill $150,000; Prepaid Insurance $12,000; Patents $220,000;...
-
This assignment is focused on project selection and the underlying factors used to make this determination. You will need to use the readings/videos, the previous learning modules, along with some...
-
Calculate the equilibrium composition of the two liquid phases in a binary mixture of methyl diethylamine (a) and water (b) at 1 bar and 20C. Solve this problem in two ways; by graphical methods and...
-
Repeat Example 8.15 using the PengRobinson equation of state. Compare your answer to the reported measured value of P = 69.1 bar and y1 = 0.95. Example 8.15 Using the equation of state method,...
-
If you were given money to invest in either a commercial bank or a mutual fund, explain which one (commercial bank or mutual fund) you would invest in or would you invest in both and why based on the...
-
From a friend who is Human Resources Manager for a law fi rm: I need your help. Our partners are contemplating the introduction of 360-degree appraisal, and have asked my advice about the benefi ts...
-
Mountaintop golf course is planning for the coming season. Investors would like to earn a 12% return on the company's $50,000,000 of assets. The company primarily incurs fixed costs to groom the...
-
Sometimes, lenders allow or require a downpayment before they extend you the loan. What would be the advantage to the lender? What would be the advantage to the borrower?!
-
Daphne bought a house ten years ago for $245,000.The real estate market has had it ups and downs since then. Daphne read a story in todays newspaper that says that during the past decade, houses in...
-
Integrate manually JxIn(2x) dx =
-
Which five issues should be resolved before a team can collaborate productively?
-
Velshi Printers has contracts to complete weekly supplements required by fortysix customers. For the year 2018, manufacturing overhead cost estimates total $600,000 for an annual production capacity...
-
Show the hybridization at each of the atoms, except H, in these molecules. Indicate the type of each designated bond and the orbital's that are overlapping to form it. : a) H_C_C,H 4 (both) : ...
-
Circle the conjugated pi bonds, if any, in the following compounds? c) CH,CH=CH-C=N b) CH3-CH=CH-C-CH3 d) H-C=C-CH=CH-C-H La e) CH-CH=CH-CH-C-CH3
-
What is the hybridization at the indicated atoms in these compounds? a) CHCH=CH_NHCH3 12 3 c) CH,=CH0CH, 1 2 3 4 5 0: b) CHC0CH, 1 d) 2 1 NH
-
How to prepare for FLIGBY simulation and perform well in the first take
-
Fixed costs: eBook Tiger Equipment Inc., a manufacturer of construction equipment, prepared the following factory overhead cost budget for the Welding Department for May of the current year. The...
-
Complete this question by entering your answers in the tabs below. Req A Req B and C Record the transactions in the accounting equation for Year 2. Record the amounts of revenue, expense, and...

Study smarter with the SolutionInn App


