Assign an oxidation number to each atom and identify the oxidizing agent and reducing agent: 3 Na2SO3
Question:
Assign an oxidation number to each atom and identify the oxidizing agent and reducing agent:
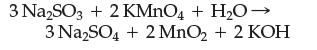
Transcribed Image Text:
3 Na2SO3 + 2 KMnO4 + H₂O → 3 Na₂SO4 + 2 MnO₂ + 2 KOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
3 NaSO 2 MnO22 KOH...View the full answer

Answered By

Gauri Hendre
I worked as EI educator for Eduphy India YT channel. I gave online tutorials to the students who were living in the villages and wanted to study much more and were preparing for NEET, TET. I gave tutions for topics in Biotechnology. I am currently working as a tutor on course hero for the biochemistry, microbiology, biology, cell biology, genetics subjects. I worked as a project intern in BAIF where did analysis on diseases mainly genetic disorders in the bovine. I worked as a trainee in serum institute of India and Vasantdada sugar institute. I am working as a writer on Quora partner program from 2019. I writing on the topics on social health issues including current COVID-19 pandemic, different concepts in science discipline. I learned foreign languages such as german and french upto A1 level. I attended different conferences in the science discipline and did trainings in cognitive skills and personality development skills from Lila Poonawalla foundation. I have been the member of Lila poonawalla foundation since 2017. Even I acquired the skills like Excel spreadsheet, MS Office, MS Powerpoint and Data entry.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
1A) Assign an oxidation number to each atom in thereactants. Na2S(aq)+NiCl2(aq)?2NaCl(aq)+NiS(s) 1B) Assign an oxidation number to each atom in theproducts. Na2S(aq)+NiCl2(aq)?2NaCl(aq)+NiS(s) 1C) Is...
-
The reaction Cu 2+ + I 2 Cu + + 2I is not possible as written. Assign an oxidation number to each atom and explain what is wrong with this reaction.
-
Consider the redox reaction (a) Identify the oxidizing agent on the left side of the reaction and write a balanced oxidation half-reaction. (b) Identify the reducing agent of the left side of the...
-
AutoSave Off File C30 PivotTable Recommended Table Pivot Tables 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 A Enter Home Insert Tables H Price Par Value B X Coupon Rate Payment Frequency...
-
The net income reported on the income statement for the current year was $210,000. Depreciation recorded on equipment and a building amounted to $62,500 for the year. Balances of the current asset...
-
Use www.congress.gov or another reliable website to find a proposal for a carbon tax or a financial transactions tax. Draft a summary of the tax, and analyze it against five of the principles of good...
-
Should Paula resign? If she submits her resignation, should David accept it, or should he ask her to continue in her position at the Texas Plant? What is best for the Texas Plant? What is best for...
-
Walshs Juice Company produces three products from unprocessed grape juicebottled juice, frozen juice concentrate, and jelly. It purchases grape juice from three vineyards near the Great Lakes. The...
-
Why the ABC brings the greatest benefits in a larger corporation? Give the reason.
-
The spontaneous redox reaction Mn + Cd 2+ Mn 2+ + Cd takes place in a battery. (a) What is the oxidizing agent? (b) What is the reducing agent? (c) Which metal is the cathode? (d) Which metal is the...
-
Assign an oxidation state to each carbon in: (a) H 3 CCH 3 (b) H 2 CCH 2 (c) HCCH
-
A sample of 100 observations produces the following sample data: y 1 = 1, y 2 = 2, y' 1 y 1 = 150, y 2 y 2 = 550, y 1 y 2 = 260. The underlying bivariate regression model is y 1 = + 1 , y 2 = + 2...
-
The answer above is NOT correct. The value of (2x + 1)(x + x)dx is
-
Review the resource on organizational theory. Explore the various theories and select one to use for this Discussion. Consider the strengths and limitations of the chosen theory. Compose an analysis...
-
How do the locations of Australian department store Myer affect the ability of the other factors of the operating model canvas (suppliers, organization, processes, and information/management systems)...
-
Critical Reading Review: The Exclusion of Latinos from American Media and History Books Read the article. After reading the article, answer the following questions: 1. What purpose do you think the...
-
1. How does the proposed market segment of residential contracts differ from Smith Electric's current market segment? 2.What does a SWOT analysis tell us about Smith Electric's ability to enter a...
-
Let L be a linear operator on R1 and let a = L(l). Show that L(x) = ax for all .v R1
-
The figure shows six containers, each of which is filled from the top. Assume that water is poured into the containers at a constant rate and each container is filled in 10 seconds. Assume also that...
-
Assuming that the switch in Fig. 7.87 has been in position A for a long time and is moved to position B at t = 0, Then at t = 1 second, the switch moves from B to C. Find v C (t) for t ¥ 0. 10 )...
-
Select a pump from the sample catalogue within the PIPE-FLO demo software to run a system that pumps water at 30C from a reservoir up into a storage tower, 20 m higher, at a rate of 1800 L/min....
-
If we assume that a given pump requires 7.50 ft of NPSH when operating at 3500 rpm, what would be the NPSH required at 2850 rpm?
-
Comfort Golf Products is considering whether to upgrade its equipment Managers are considering two options. Equipment manufactured by Stenback Inc. costs $1,000,000 and will last five years and have...
-
Weaver Corporation had the following stock issued and outstanding at January 1, Year 1: 71,000 shares of $10 par common stock. 8,500 shares of $60 par, 6 percent, noncumulative preferred stock. On...
-
Read the following case and then answer questions On 1 January 2016 a company purchased a machine at a cost of $3,000. Its useful life is estimated to be 10 years and then it has a residual value of...

Study smarter with the SolutionInn App


