Consider the following reaction. Describe it in terms of which bonds must be broken and which bonds
Question:
Consider the following reaction. Describe it in terms of which bonds must be broken and which bonds must be formed.
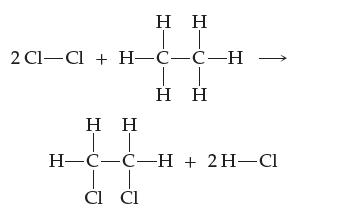
Transcribed Image Text:
Η Η TT 2Cl—Cl + H-C-C-H Η Η Η Η IT H=C=C-H + 2H-Cl T CI CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
H H H CH H H Ethane Explanation 21 Chlorine molecule H H H H ...View the full answer

Answered By

Rehab Rahim
I am well versed in communicating and teaching in areas of all business subjects. I have helped many students in different ways from answering answers to writing their academic papers.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Describe the following reaction in terms of which bonds must be broken and which bonds must be formed: HC=CH + 30 2 COz+2 H,O
-
Is it likely that a single collision leads to the breaking of all the bonds that you named in Problem 13.27? Explain your answer. Data from Problem 13.27 Describe the following reaction in terms of...
-
Consider the following reaction. Use labeled arrows to indicate which bonds must be broken and which bonds must be formed. H-CC-0-H + Cl HC-C-0-C-C-H + HCl H C-C-H
-
On July 31, 2023, Magnuss Muffins paid $18,000 for business insurance for the next year. Record the entries for the purchase of the insurance by recording it as an expense and then making a year-end...
-
Hanno Legal Services provides legal advice to clients. The following data apply to the first six months of operation: .:. Required a. What is the average service revenue per hour for the six-month...
-
Torrent plcs income statement for the year ended 31 December 2010 and the statements of financial position as at 31 December 2009 and 2010 are as follows: During 2010, the business spent £67...
-
Estimation of urban population by means of satellite images. Refer to the Geographical Analysis (Jan. 2007) study that demonstrated the use of satellite image maps for estimating urban population,...
-
1. Identify some of the problems likely to occur in a boundaryless organization such as Newskool Grooves. What are the advantages of boundaryless organizations? 2. Consider some of the cultural...
-
Thumbs up to correct answers!!!! formulas would be appreciated! Current Attempt in Progress Comparative financial statements for Ivanhoe and Bridgeport Ltd. are shown below. IVANHOE AND BRIDGEPORT...
-
Under what circumstances (if any) does it not take energy to break a chemical bond?
-
Recall the reaction to make the superconductor described at the beginning of this chapter. What could be done to show that the product of the chemical reaction is truly a new compound and not just a...
-
Why do you think the designers of Linux made it impossible for a process to send a signal to another process that is not in its process group?
-
Required: Prepare the supporting schedules for your portfolio for presentation to Mandla the supervisor and senior administrator. The schedules for the portfolio need to cover the following: Part A...
-
Write a program that will predict the size of a population of organisms. The program should ask for the starting number of organisms, their average daily population increase (as a percentage), and...
-
Management is keen to reduce inventory levels for materials as well and closing inventories are to be much lower. Expected levels are shown below: Material M1 Material M2 Material M3 2,200 kg 1,300...
-
How do you calculate incremental cost for the following: Complying with the Clean Air Act Amendments will be costly. There are three main options for complying with the Clean Air Act: analyze the...
-
How do I journalize this transaction? Mountain Swirl Ice Cream purchased and took delivery of one ice cream machine for $7,500. Record the sale and the cost of the sale. Markup is 150% of cost....
-
How many bonding interactions are there in the 1 and 2 molecular orbitals of the following compounds? a. 1,3-butadiene b. 1,3,5,7-octatetraene
-
From the choice of simple moving average, exponential smoothing, and linear regression analysis, which forecasting technique would you consider the most accurate? Why? please write it in word...
-
Natural light of irradiance I i is incident normally on an HN-32 polarizer. (a) How much light emerges from it? (b) A second identical polarizer is placed parallel to and behind the first. How much...
-
Imagine four HN-32 Polaroids one behind the other with their transmission axes all parallel. If the irradiance of natural light incident on the first filter is I i , what is the transmitted...
-
The irradiance of a beam of natural light is 400 W/m 2 . It impinges on the first of two consecutive ideal linear polarizers whose transmission axes are 40.0 apart. How much light emerges from the...
-
Show that the convexity for a zero coupon bond with m payments per year is (m) n(n + -)(1+ m m
-
Abdul Canarte , a Central Bank economist, noticed that the total group purchasing basket of goods (CPI) has gone from $149,740.00 to $344,460.00 in 8 years. With monthly compounding, what is the...
-
ABC Corporation expects sales next year to be $50,000,000. Inventory and accounts receivable (combined) will increase $8,000,000 to accommodate this sales level. The company has a profit margin of 6...

Study smarter with the SolutionInn App


