Does the following reaction-energy profile represent an endothermic or exothermic reaction in the forward direction? In the
Question:
Does the following reaction-energy profile represent an endothermic or exothermic reaction in the forward direction? In the reverse direction?
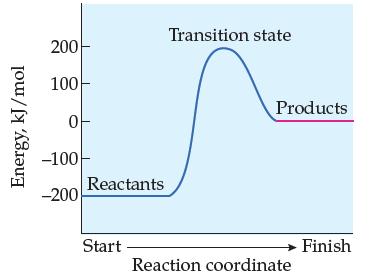
Transcribed Image Text:
Energy, kJ/mol 200 100- 0 -100 -200 Reactants Start Transition state Products Reaction coordinate Finish
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The reactionenergy profile represents an endothermic reaction in the forward direction and an exoth...View the full answer

Answered By

Muhammad Haroon
More than 3 years experience in teaching undergraduate and graduate level courses which includes Object Oriented Programming, Data Structures, Algorithms, Database Systems, Theory of Automata, Theory of Computation, Database Administration, Web Technologies etc.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
What is the activation energy for the reverse reaction in Problem 13.95? Data from Problem 13.95 Does the following reaction-energy profile represent an endothermic or exothermic reaction in the...
-
What is the activation energy for the forward reaction in Problem 13.95? Data from Problem 13.95 Does the following reaction-energy profile represent an endothermic or exothermic reaction in the...
-
What is E forward rxn for the reaction in Problem 13.95? What is E reverse rxn ? Data from Problem 13.95 Does the following reaction-energy profile represent an endothermic or exothermic reaction in...
-
True or False? Azure files can be accessed from anywhere in the world using a URL that points to the file. True False
-
Inferring Year-End Adjustments, Computing Earnings per Share and Net Profit Margin, and Recording Closing Entries Ramirez Company is completing the information processing cycle at its fiscal...
-
The H.R. Pickett Corp. has $500,000 of debt outstanding, and it pays an annual interest rate of 10%. Its annual sales are $2 million, its average tax rate is 30%, and its net profit margin is 5%....
-
Can nuclei of the same element have different values of \(Z\) ? Of \(N\) ? Of \(A\) ? Can nuclei of different elements have the same values of \(Z\) ? Of \(N\) ? Of \(A\) ?
-
There is often more than one way to improve a performance measure. Unfortunately, some of the actions taken by managers to make their performance look better may actually harm the organization. For...
-
Chuck, a single taxpayer, earns $75,800 in taxable income and $10,900 in interest from an investment in City of Heflin bonds. (Use the U.S. tax rate schedule.) Required: a. If Chuck earns an...
-
From the following reaction-energy profiles, determine whether reactions A and B are exothermic or endothermic in the forward direction: Energy, kJ/mol 300 200 100 0 -100 -200 B Reactants Start...
-
In a kinetic study of the reaction the following rate data were obtained. Write a rate law complete with proper values for the orders. What is the overall order of the reaction? 2 C10(aq)+ 2 OH (aq) ...
-
Jan Voda, M.D., a resident of Oklahoma City, Oklahoma, owns three U.S. patents related to guiding catheters for use in interventional cardiology, as well as corresponding foreign patents issued by...
-
What idea, concept or issue stood out in Medicare and Medicaid If you are aspire to be a health services manager, how might you use Medicare and Medicaid in your work?
-
Harvard Business Publishing Project Management Simulation: Scope, Resources, and Schedule Simulation Scenarios A, B,C, D, E, F & G Student experience personal and group? what was your impression?...
-
Describe the purpose, procedures strengths and limitations of electronic or computer based training methods.
-
Summarize a specific company's situation that involved business continuity and disaster recovery planning. Explain the impact the situation had on the organization
-
Why is it important for HR and compensation professionals to learn about compensation practices in other parts of the world? Include a rationale for your answer.
-
Upon returning from lunch, you find the following message on your voice mail: This is Jarrett Ogilvie. I'm not one of your clients, but I need some advice. I received a statement in the mail from the...
-
Use integration by parts to evaluate the following. Check your answer by taking the derivative. x2e-xdx
-
A baseball is thrown with an initial velocity of magnitude v 0 at an angle of 60 with respect to the horizontal (x) direction. At the same time, a second ball is thrown with the same initial speed at...
-
A high jumper can run horizontally with a top speed of 10.0 m/s. (a) If he can convert this velocity to the vertical direction when he leaves the ground, what is the theoretical limit on the height...
-
A bullet is fired from a rifle with speed v 0 at an angle θ with respect to the horizontal axis (Fig. P4.32) from a cliff that is a height h above the ground below. (a) Calculate the...
-
Many successful technological innovations originated in small start-up organizations, while others originated in large, well-established organizations. Successful technological innovations can occur...
-
what is one benefit and a drawback of The central bank finances itself from the earnings on its assets and turns the balance over to the government?
-
Based on your Daily Spending Diary, describe actions that you might take to identify and achieve various financial and investment goals.

Study smarter with the SolutionInn App


