What is the activation energy for the reverse reaction in Problem 13.95? Data from Problem 13.95 Does
Question:
What is the activation energy for the reverse reaction in Problem 13.95?
Data from Problem 13.95
Does the following reaction-energy profile represent an endothermic or exothermic reaction in the forward direction? In the reverse direction?
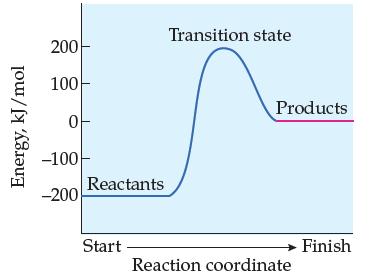
Transcribed Image Text:
Energy, kJ/mol 200 100- 0 -100 -200 Reactants Start Transition state Products Reaction coordinate Finish
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The activation energy for the reverse reaction in Problem 1395 is 260 kJmol This is because the reve...View the full answer

Answered By

Abigael martinez
I have been a tutor for over 3 years and have had the opportunity to work with students of all ages and backgrounds. I have a strong belief that all students have the ability to learn and succeed if given the right tools and support. I am patient and adaptable, and I take the time to get to know each student's individual learning style in order to best support their needs. I am confident in my ability to help students improve their grades and reach their academic goals.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
What is the activation energy for the forward reaction in Problem 13.95? Data from Problem 13.95 Does the following reaction-energy profile represent an endothermic or exothermic reaction in the...
-
What is E forward rxn for the reaction in Problem 13.95? What is E reverse rxn ? Data from Problem 13.95 Does the following reaction-energy profile represent an endothermic or exothermic reaction in...
-
Does the following reaction-energy profile represent an endothermic or exothermic reaction in the forward direction? In the reverse direction? Energy, kJ/mol 200 100- 0 -100 -200 Reactants Start...
-
Suppose an economy is at full-employment equilibrium at a GDP of $25 billion and investment declines by $4 billion. According to Keynes, this economy will: OA. Quickly self adjust back to full...
-
Recording Transactions (Including Adjusting and Closing Entries), Preparing a Complete Set of Financial Statements, and Performing Ratio Analysis Josh and Kelly McKay began operations of their...
-
Explain briefly the tax treatment of (1) interest and dividends paid, (2) interest earned and dividends received, (3) capital gains, and (4) tax loss carry-back and carry-forward. How might each of...
-
An atom has four energy levels. How many spectral lines are seen in its emission spectrum? In its absorption spectrum? How many absorption lines?
-
The following crosstabulation shows household income by educational level of the head of household Statistical Abstract of the United States, 2008). a. Develop a joint probability table. b. What is...
-
Roman Company was established a few years ago. The company operates all over the beautiful Pacific Northwest. Roman Company has just completed its current accounting period for the year. Roman...
-
From the following reaction-energy profiles, determine whether reactions A and B are exothermic or endothermic in the forward direction: Energy, kJ/mol 300 200 100 0 -100 -200 B Reactants Start...
-
In a kinetic study of the reaction the following rate data were obtained. Write a rate law complete with proper values for the orders. What is the overall order of the reaction? 2 C10(aq)+ 2 OH (aq) ...
-
Let each subgroup consist of the 6 values within a year. Construct an x chart and determine whether the process mean is within statistical control. If it is not, identify which of the three...
-
Explain why the process of critical reflection in relation to diversity is important asaneducator.
-
Select 3 different Warehouse Coordinator Job Position for multinational company specializing in Global Sourcing from online jobsites (Position need to be completely presented as advertised with...
-
What legal and ethical considerations should a counsellor be aware of when working with self-harm and suicidal behaviours?
-
Public Sector unions now account for more than half of union members, while the private sector accounts for less than half. Research this topic using online professional articles and give your...
-
What does the company Coca-Cola state about its view on ethics?
-
Erica and Raphael are divorced during the current year. Because Erica made millions in the record industry while Raphael served as the homemaker and primary caretaker of baby Dexter, Erica agrees to...
-
Solve for the equilibria of the following discrete-time dynamical systems Pr pt+1 = Pr+2.0(I-Pr)
-
Two children pull a sled of mass 15 kg along a friction less surface as shown in Figure P4.43. (a) Find the magnitude and direction of the sleds acceleration. (b) How long does it take the sled to...
-
A 35-m-wide river flows in a straight line (the x direction) with a speed of 0.25 m/s. A boat is rowed such that it travels directly across the river (along y). If the boat takes exactly 4 minutes to...
-
Round trip. An airplane flies from Chicago to New Orleans (a distance of 1500 km) in the morning and then immediately returns to Chicago. The airplanes speed relative to the air is 250 m/s. The wind...
-
Product Profitability Analysis Galaxy Sports Inc. manufactures and sells two styles of All Terrain Vehicles (ATVS), the Conquistador and Hurricane, from a single manufacturing facility. The...
-
what extent does socialization influence political attitudes, ideologies, and participation within democratic societies?
-
Money Corp. is looking to invest $115,317 in either a State of California bond or a corporate bond. If the corporate bond yields 4%, what rate would the State of California bond need to pay to yield...

Study smarter with the SolutionInn App


