What is the activation energy for the forward reaction in Problem 13.95? Data from Problem 13.95 Does
Question:
What is the activation energy for the forward reaction in Problem 13.95?
Data from Problem 13.95
Does the following reaction-energy profile represent an endothermic or exothermic reaction in the forward direction? In the reverse direction?
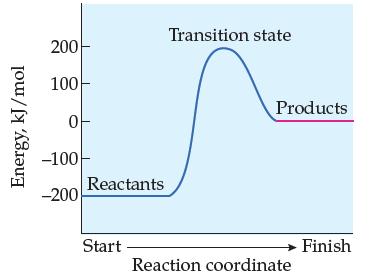
Transcribed Image Text:
Energy, kJ/mol 200 100- 0 -100 -200 Reactants Start Transition state Products Reaction coordinate Finish
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The activation energy for the forward reaction in Problem 1395 is the energy difference between ...View the full answer

Answered By

Shadrack Mulunga
I am a Biochemistry by profession. However, I have explored different fields of study. My quest to explore new fields has helped me gain new knowledge and skills in Business, clinical psychology, sociology, organizational behavior and general management, and Project Management. I count my expertise in Project management, in particular, creation of Work Break Down Structure (WBS) and use of Microsoft Project software as one of my greatest achievement in Freelancing industry. I have helped thousands of BSC and MSC students to complete their projects on time and cost-effectively using the MS Project tool. Generally, I find happiness in translating my knowledge and expertise to success of my clients. So far, i have helped thousands of students to not only complete their projects in time but also receive high grades in their respective courses. Quality and timely delivery are the two key aspects that define my work. All those who hired my services always come back for my service. If you hire my services today, you will surely return for more. Try me today!
5.00+
154+ Reviews
289+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
What is the activation energy for the reverse reaction in Problem 13.95? Data from Problem 13.95 Does the following reaction-energy profile represent an endothermic or exothermic reaction in the...
-
What is E forward rxn for the reaction in Problem 13.95? What is E reverse rxn ? Data from Problem 13.95 Does the following reaction-energy profile represent an endothermic or exothermic reaction in...
-
Does the following reaction-energy profile represent an endothermic or exothermic reaction in the forward direction? In the reverse direction? Energy, kJ/mol 200 100- 0 -100 -200 Reactants Start...
-
1) Explain what creates value and identify if there are aspects that do not create value. (Please include the citation and references) 2)What is the strength of Hyatt Hotel's Loyalty 3)What makes you...
-
Recording Transactions (Including Adjusting and Closing Entries), Preparing a Complete Set of Financial Statements, and Performing Ratio Analysis Brothers Mike and Tim Hargen began operations of...
-
Indicate the effects of the transactions listed in the following table on total current assets, current ratio, and net income. Use (+) to indicate an increase, (-) to indicate a decrease, and (0) to...
-
A photon with a wave-length of 410 nm has energy E photon = 3.0 eV. Do you expect to see a spectral line with = 410 nm in the emission spectrum of the atom represented by this energy-level diagram?...
-
Nellie works for a large Fortune 500 company. She heads the information systems department and works closely with the accounting department. The company works with many associates. They have many...
-
When running the program given, it is supposed to give the output bewlow. However, the program contains erros that prevent it from compiling and/or running. Correct the program so that it works...
-
From the following reaction-energy profiles, determine whether reactions A and B are exothermic or endothermic in the forward direction: Energy, kJ/mol 300 200 100 0 -100 -200 B Reactants Start...
-
In a kinetic study of the reaction the following rate data were obtained. Write a rate law complete with proper values for the orders. What is the overall order of the reaction? 2 C10(aq)+ 2 OH (aq) ...
-
In 2003, President Bush proposed a change in the tax law that would have eliminated the tax on dividends received by stockholder. The same proposal also would have increased the basis of stocks by...
-
Does Coca-Cola company published view on ethical standards conform to or differ from its practices of individualism view, moral rights view, justice view, and utilitarian view versus their practiced...
-
A thermocouple made of Chromel/alumel is placed in an oven and connected to a computer DAQ system. The Junction box temperature is independently measured to be 25 o C. The thermo -couple voltage is...
-
Two objects, each having a mass of 2.3 x 10 8 kg, are separated by a distance of 3.0 x 10 3 m. What is the gravitational force of attraction between them?
-
For the common-base network of Fig. 4.138 a. Using the information provided determine the value of Ro b. Find the currents I and I. c. Determine the voltages VBC and VCE 14 V RC Ve=8V H 4Vo 8=90 RE...
-
The salt-water battery, which is currently under development in the USA and Australia, is claimed to be an attractive alternative to the lead-acid battery for utility load-leveling and peak shaving...
-
Will and Janine are divorced during the current year. Will is to have custody of their two children and will receive their house as part of the divorce settlement. The house, which Will and Janine...
-
Suppose that fraction used = / 1.0 + 0.1Mt. for some parameter 1. Write the discrete-time dynamical system and solve for the equilibrium. Sketch a graph of the equilibrium as a function of ....
-
An airplane flies from Boston to San Francisco (a distance of 5000 km) in the morning and then immediately returns to Boston. The airplanes speed relative to the air is 250 m/s. The wind is blowing...
-
Consider again the airplane in Problem 38, but now suppose the wind is directed in an easterly direction. How long does it take the airplane to travel a distance between two cities that are 300 km...
-
An airplane has a velocity relative to the air of 200 m/s in a westerly direction. If the wind has a speed relative to the ground of 60 m/s directed to the north, what is the speed of the airplane...
-
You started a venture 2 years ago with $400,000 dollars and own 60% of the 500,000 shares issued. What is the pre and post money valuations under each deal offered: Alpha Ventures offers $200,000 for...
-
On April 30, the end of the first month of operations, Joplin Company prepared the following income statement, based on the absorption costing concept: Joplin Company Absorption Costing Income...
-
Based on the above creative & article write the creative brief that you feel the campaign is based on. Remember the creative brief comes BEFORE the campaign is develop and needs to be worded as so....

Study smarter with the SolutionInn App


