In Reaction 15.7, water is shown acting as a base. How is water behaving in the following
Question:
In Reaction 15.7, water is shown acting as a base. How is water behaving in the following reaction?
![]()
Explain your answer.
Reaction 15.7
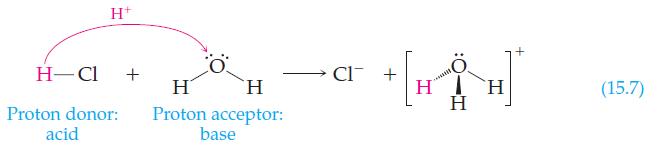
Transcribed Image Text:
NH₂ + H₂O → NH3 + OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Answer In the reaction NH 2 H 2 O NH 3 OH water is acting as an acid This can be understood through ...View the full answer

Answered By

Mamba Dedan
I am a computer scientist specializing in database management, OS, networking, and software development. I have a knack for database work, Operating systems, networking, and programming, I can give you the best solution on this without any hesitation. I have a knack in software development with key skills in UML diagrams, storyboarding, code development, software testing and implementation on several platforms.
4.90+
49+ Reviews
119+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
In Chapter 10, we will see that an acetylide ion (formed by treatment of acetylene with a strong base) can serve as a nucleophile in an S N 2 reaction: This reaction provides a useful method for...
-
In this part of the project you are to assume to have been hired to join a team serving as an internal financial analyst to THE COMPANY. Your client plans to invest in bonds and (or) stocks issued by...
-
It is important to be able to distinguish when an electron pair donor in a reaction is acting as a base or as a nucleophile. The following reactions are elementary reactions. In each case, determine...
-
Assume that the average talk time on an Apple iPhone is 20 hours and that this time follows the exponential probability distribution. What is the probability that a randomly selected iPhone will...
-
The financial statements for Highland Publications Corporation included the following selected information: Common stock $1,600,000 Retained earnings $900,000 Net income $1,000,000 Shares issued...
-
List and briefly explain the special features usually associated with preferred stock.
-
Amanda Forbes was hired as a nail technician by Showmann, Inc., in 2011. In 2017, Forbes attended a work-related holiday party where Showmann distributed raffle tickets to employees. One of the...
-
Weighted-average method. Ashworth Handcraft is a manufacturer of picture frames for large retailers. Every picture frame passes through two departments: the assembly department and the finishing...
-
The N excess/deficit factor for 100 kg of an organic material that contained 60% C (carbon) and 0.5% N (nitrogen) would likely be (assuming that 35% of the carbon is metabolized by microorganisms and...
-
(a) How does a weak acid in a buffer eliminate added OH ions? To what does it convert these ions? What happens to the weak acid? (b) How does a weak base in a buffer eliminate added H 3 O + ions? To...
-
Draw three beakers as shown in WorkPatch 15.3, but change the labels to make them reflect the reaction mentioned in Practice Problem 15.9. Then list all species present in each beaker. Data from...
-
The lithium atom has a nuclear charge of +3e. Find the energy required to remove the third electron from a lithium atom that has already lost two of its electrons. Assume the third electron to be...
-
Your firm has tangible assets of $101.6 million. You are planning to acquire a firm that is half your firm's size. You have bonds with a merger & acquisition covenant that requires the combined firm...
-
Estimated production for next year = 2,60,000 units. Estimated no. of workers needed (on the basis of ratio-trend of 1: 400). What is the manpower required for next year? Use ratio analysis.The...
-
6. Express as a single, simplified logarithm. 1 (logs 10+2log53)
-
just finished the business plan of his start-up company. According to the projections he carried-out, the initial investment is 1,500,000 SAR (assume that we are in the beginning of 2023), which will...
-
1. You have $100,000 to invest in a Stock S and Stock T portfolio. The following table contains the essential information for the portfolio: Economic State Boom Normal Recession Weight for each stock...
-
On August 10, 2010, Black, Inc. acquired an office building as a result of a like-kind exchange. Black had given up a factory building that it had owned for 18 months as part of the like-kind...
-
Consider the discrete group G of order 8 that has the following Cayley diagram e If we have the sequence of operations: fcagec, which of the options represents the reduction of the sequence to a...
-
The structural assembly supports the loading shown. Draw the moment diagrams for each of the beams. Take I = 100(10 6 ) mm 4 for the beams and A = 200 mm 2 for the tie rod. All members are made of...
-
The contilevered beam is supported at one end by a 1/2 in.-diameter suspender rod AC and fixed at the other end B. Determine the force in the rod due to a uniform loading of 4 k/ft. E = 29(10 3 ) ksi...
-
The beam AB has a moment of inertia I = 475 in 4 and rests on the smooth supports at its ends. A 0.75-in-diameter rod CD is welded to the center of the beam and to the fixed support at D. If the...
-
Multiple Choice Question Sofia sees a customer at the grocery store shout at a cashier. "That customer is a jerk," thinks Sofia, but she doesn't know that the cashier has just been caught trying to...
-
Topic 8 Midterm Start Date & Time Feb 22, 2024, 12:00 AM Due Date & Time Feb 26, 2024, 11:59 PM Time Limit 1 hr 15 mins Status Upcoming Assessment Traits Timed Assessment Description Complete the...
-
If sales are over $1000, the bonus is 10%. If sales are over $500, the bonus is 5%. Otherwise, there is no bonus. Which formula calculates the correct bonus percentage? =IF(A1 > 1000, "10%", "5%")...

Study smarter with the SolutionInn App


