The following is a two-step mechanism for how chlorine atoms in the upper atmosphere react with and
Question:
The following is a two-step mechanism for how chlorine atoms in the upper atmosphere react with and decompose ozone.
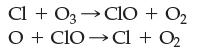
According to the mechanism, which is the intermediate and which is the catalyst? Justify your choices.
Transcribed Image Text:
Cl + O3 →CIO 03 + O₂ OCIO Cl + O₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
According to the image the chloride atom Cl is the catalyst in the twostep mechanism for how chlorin...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
A proposed two-step mechanism for the destruction of ozone in the upper atmosphere is a. What is the overall balanced equation for the ozone destruction reaction? b. Which species is a catalyst? c....
-
You are a contractor that has been asked to build an apartment complex. The associated AON Network diagram and the Time/Cost table are provided below. (Based on the table, you could crash activity B...
-
The following is a questionnaire to be completed by customers of the Hilltop Smoked Meat Co. Restaurant. Build a codebook that might be used to transfer raw answers from completed questionnaires to a...
-
Norma received a deficiency letter from the Internal Revenue Service. She appealed to the Independent Office of Appeals, and her appeal was denied. Now she wants to take the government to court....
-
What is a journal entry?
-
Stocks X and Y have the following probability distributions of expected future returns: a. Calculate the expected rate of return, rY, for Stock Y (rX = 12%).b. Calculate the standard deviation of...
-
Consider the gold isotope \({ }^{197} \mathrm{Au}\). a. How many electrons, protons, and neutrons are in a neutral \({ }^{197} \mathrm{Au}\) atom? b. The gold nucleus has a diameter of \(14.0...
-
Dodson Co. is planning to finance an expansion of its operations by borrowing $120,000. City Bank has agreed to loan Dodson the funds. Dodson has two repayment options: (1) to issue anote with the...
-
It's amazing to see how far behind China is in terms of individual household income compared to other countries with similar economic standing. Do you think it's due to its sheer population size? Or...
-
A student claims that an endothermic reaction will always have a higher activation energy than an exothermic reaction, because an endothermic reaction ends up with the products at a higher energy...
-
Consider the decomposition of ozone (O 3 ) to oxygen (O 2 ). The rate law for this reaction is: Rate = k[O 3 ] 2 /[O 2 ]. How is the rate of this reaction affected by the concentration of oxygen?...
-
Outline media (including social media) research issues that are relevant for today's marketing communicator.
-
In today's business world, communication channels are increasing rapidly, and the demand for connectivity between employees and organizational leaders is also growing, which has further heightened...
-
How does Amazon's communication system support upward, downward, and horizontal communication? How does Amazon's communication system reflect the implementation of the interpersonal communication...
-
Please provide critique of the article "Using Real-World Examples to Enhance the Relevance of theIntroductory Statistics Course" by Friedman, Friedman and Amoo. Link...
-
Define and describe the processes of project communication management according to the extract above. Describe the meaning of communication to implemented projects and what the project managers must...
-
Bob is doing a quality test on machine bolts. He tested 24 bolts that he randomly picked and found that 5 were defective. base on bobs results, what is the expected number of bolts that will be...
-
Gilbert got married this year. Because he couldnt afford a wedding reception, his employer gave him the $5,000 he needed to pay for it. Identify the tax issue(s) posed by the facts presented....
-
In the operation of an automated production line with storage buffers, what does it mean if a buffer is nearly always empty or nearly always full?
-
A rocket-powered sled of mass 3500 kg travels on a level snow-covered surface with an acceleration of +3.5 m/s 2 (Fig. P3.7). What are the magnitude and direction of the force on the sled? Figure...
-
Two balls are thrown from a tall bridge. One is thrown upward with an initial velocity +v 0 , while the other is thrown downward with an initial velocity -v 0 . Which one has the greater speed just...
-
Your car has a dead battery. It is initially at rest, and you push it along a level road with a force of 120 N, finding that it reaches a velocity of 2.0 m/s in 50 s. What is the mass of the car?...
-
What are the challenges and limitations of applying conflict theory in empirical research and social policy, particularly in terms of addressing complexities and nuances in power dynamics and social...
-
Given P(x)=325 + 2x + 85x3 + 42x2 (as a product of linear factors). 8282-1080, and that 6i is a zero, write P in factored form
-
The polynomial 3x^(2) - 10x+8 has a factor of 3x-4. What is the other factor of 3x^(2) - 10x + 8?

Study smarter with the SolutionInn App


