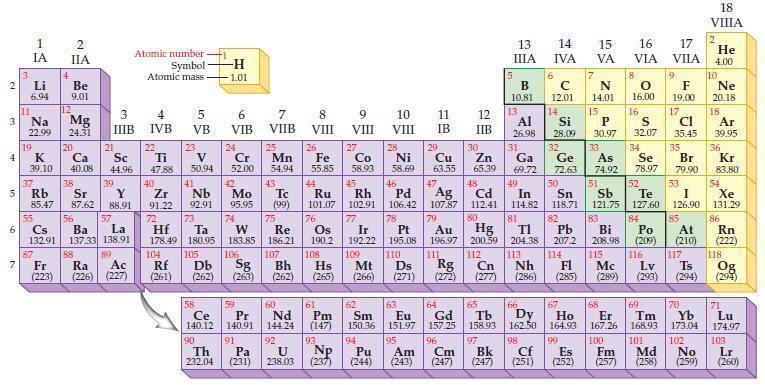
Predict the ionic charge for an aluminum ion, Al ?+ , based on the position of the
Question:
Predict the ionic charge for an aluminum ion, Al?+, based on the position of the element in the periodic table.
Periodic Table:
Transcribed Image Text:
2 3 4 15 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 Rb R 4 87 2 IIA Be 9.01 12 Mg 24.31 K Ca Sc 39.10 40.08 44.96 20 38 21 3 IIIB 39 Sr Y 85.47 87.62 88.91 57 55 56 La Cs Ba 132.91 137.33 138.91 89 88 Fr Ac Ra (223) (226) (227) Atomic number Symbol Atomic mass 4 IVB 22 Ti 47.88 40 Zr 2 91.22 72 5 VB 104 23 V 50.94 41 73 Hf Ta 178.49 180.95 105 Rf Db (261) (262) -H -1.01 6 VIB Nb Mo 92.91 95.95 90 24 Cr 52.00 42 74 106 Sg (263) 58 Pr Ce 140.12 140.91 91 59 7 VIIB Th Pa 232.04 (231) 25 W Re 183.85 186.21 Mn 54.94 43. Tc (99) 75 107 Bh (262) 60 Nd 144.24 92 U 238.03 8 VIII 26 Fe 55.85 76 Os 190.2 61 Pm (147) 93 NP 9 VIII (237) 27 10 VIII 11 IB 28 29 Co Zn Ni Cu 58.93 58.69 63.55 65.39 12 IIB 47 30 44 49 45 46 48 Ru Rh Pd Cd In 101.07 102.91 106.42 107.87 112.41 114.82 Ag 77 78 80 79 Au Hg 196.97 200.59 111 112 108 Ir Pt 192.22 195.08 109 110 Hs Mt Ds Rg Cn (265) (266) (271) (272) (277) 5 13 ΠΙΑ B 10.81 13 6 Ga 69.72 14 IVA с 12.01 Al Si 26.98 28.09 32 31 Ge 72.63 14 15 16 17 VA VIA VILA 66 67 62 63 64 65 Sm Eu Gd Tb Dy Ho 150.36 151.97 157.25 158.93 162.50 164.93 99 94 95 96 97 Pu Am Cm Bk (244) (243) (247) (247) 7 N 14.01 15 P 30.97 S 32.07 34 33 35 50 As Se Br 74.92 78.97 79.90 51 52 53 Sn Sb Te I 118.71 121.75 127.60 126.90 82 83 84 85 TI Pb Bi Po At 204.38 207.2 208.98 (209) (210) 113 114 115 116 117 Nh Fl Mc Lv Ts (286) (285) (289) (293) (294) 81 8 68 O 16.00 98 167.26 100 Cf Es Fm (251) (252) (257) 16 9 F 19.00 17 Cl 35.45 69 70 Er Tm Yb 168.93 173.04 101 102 Md No (258) (259) 18 VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39,95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 Lr (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Predict the ionic charge for a phosphorus ion, P ? , based on the position of the element in the periodic table. Periodic Table: 2 3 4 15 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 Rb R 4 87 2 IIA Be 9.01...
-
Predict the ionic charge for a chlorine ion, Cl ?- , based on the position of the element in the periodic table. Periodic Table: 2 3 4 5 6 7 Li 6.94 11 1 IA Na 22.99 19 37 55 4 2 IIA 87 Be 9.01 12 K...
-
Predict the ionic charge for a sodium ion, Na ?+ , based on the position of the element in the periodic table. Periodic Table: 2 3 4 5 6 7 Li 6.94 11 Na 22.99 19 1 IA K 39.10 37 55 4 2 IIA 87 Be 9.01...
-
Information sent electronically over the Internet affords little privacy. One of the reasons that cryptography is included in this text is because its importance is growing due to the increasing need...
-
Discuss the conceptual framework of accounting for federal agencies and compare it to the conceptual framework established by the GASB for state and local governments.
-
Heather Company has $10,000 to invest. One alternative will yield 10% per year, compounded annually for four years. A second alternative is to deposit the $10,000 in a bank that will pay 8% per year,...
-
Look up the FASB home page on the Internet at the following address: www.fasb.org. Find the list of technical projects that are currently on the boards agenda. Choose one of the projects that will...
-
A gas containing nitrogen, benzene, and toluene is in equilibrium with a 40 mole% benzene 60 mole% toluene liquid mixture at 100C and 10 atm. Estimate the gas-phase composition (mole fractions) using...
-
If you are given $100,000 for stock investment, how many different company's stocks will you pick to invest. How would you pick companies to make sure your portfolio is diversified?
-
What is the term for the elements that belong to Groups IAVIIIA (Groups 1, 2, and 1318)?
-
State the trends in the periodic table for decreasing metallic character. Periodic Table: 2 3 4 10 6 3 7 11 1 IA Li 6.94 Na 22.99 19 37 5 Rb K 39.10 4 87 2 IIA Fr (223) Be 9.01 12 Mg 24.31 20 38 Ca...
-
Write a filter that reads in a sequence of integers and prints the integers, removing repeated values that appear consecutively. For example, if the input is 1 2 2 1 5 1 1 7 7 7 7 1 1 1 1 1 1 1 1 1,...
-
In 1981, the election for governor of the state of New Jersey in which Tom Kean defeated Jim Florio was so close that Florio demanded a recounting of the votes. If you were Florio and you were...
-
On June 16, 2016, the worlds biggest Disneyland opened in Shanghai with a great deal of fanfare. It features a supersized 200-foot-tall castle. In comparison, the height for similar castles in...
-
A gas stations owner found that about two cars come into the station every minute. If the arrival time follows an exponential distribution, what is the probability that the next car will arrive in...
-
Find the following probabilities for the t distributions. (a) P(t> 3.078) (b) P(t < 1.943) (c) P(t > 2.492) if v = 1 if v = 6 if v = 24
-
The 80 members of a random sample of graduates of Marys Typing School indicate that their mean salary is $22,500 with a sample standard deviation of $3,100. Construct a 99 % confidence interval for...
-
The American Chamber of Commerce Researchers Association compiles cost-of-living indexes for selected metropolitan areas. Shown here are cost-of-living indexes for 25 different cities on five...
-
Nate prepares slides for his microscope. In 1 day he prepared 12 different slides. Which equation best represents y, the total number of slides Nate prepares in x days if he continues at this rate? A...
-
Albertson Corporation is a diversified company with nationwide interests in commercial real estate developments, banking, copper mining, and metal fabrication. The company has offices and operating...
-
Baden Corporation entered into a lease agreement for 10 photocopy machines for its corporate headquarters. The lease agreement qualifies as an operating lease in all terms except there is a...
-
On January 1, 2011, Perriman Company sold equipment for cash and leased it back. As seller-lessee, Perriman retained the right to substantially all of the remaining use of the equipment. The term of...
-
The former managing director of Fuji Xerox in Australia and New Zealand was paid more than AU$1 million to leave the company after it was discovered that the "inappropriate" accounting practices that...
-
Manipulation of information presented through financial reports can raise significant red flags for suspected fraud. However, it is known that information is very useful for users of financial...
-
On 1 January 2024, Tom Lee started a web design firm, Web Solutions (WS). The following are the transactions that occur during the first month of operations. 01 Jan. Tom invested $20,000 cash, and a...

Study smarter with the SolutionInn App


