State the trends in the periodic table for decreasing metallic character. Periodic Table: 2 3 4 10
Question:
State the trends in the periodic table for decreasing metallic character.
Periodic Table:
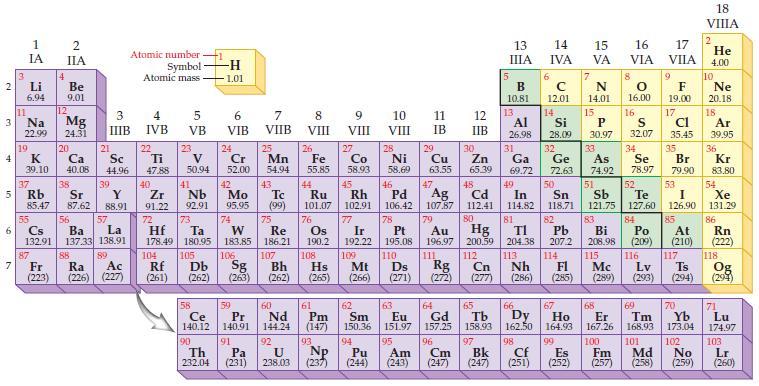
Transcribed Image Text:
2 3 4 10 6 3 7 11 1 IA Li 6.94 Na 22.99 19 37 5 Rb K 39.10 4 87 2 IIA Fr (223) Be 9.01 12 Mg 24.31 20 38 Ca Sc 40.08 44.96 21 Sr Y 85.47 87.62 88.91 55 56 La Cs Ba 132.91 137.33 138.91 88 3 4 IIIB IVB 39 57 89 Atomic number Symbol Atomic mass Ac Ra (226) (227) 22 Ti 47.88 5 VB 23 104 V 50.94 40 Nb Zr 91.22 92.91 41 105 -H 1.01 6 VIB 58 Ce 140.12 90 Th 232.04 24 Cr 52.00 42 Mo 95.95 74 72 73 W Re Hf Ta 178.49 180.95 183.85 186.21 106 Rf Db Sg (261) (262) (263) 59 Pr 140.91 91 7 VIIB Pa (231) 25 Mn 54.94 43 Tc (99) 75 107 60 Nd 144.24 8 VIII 92 26 27 Fe Co 55.85 58.93 Bh Hs (262) (265) 44 45 Ru Rh 101.07 102.91 76 Os 190.2 108 61 Pm (147) 9 VIII 93 U NP 238.03 (237) 77 Ir 192.22 109 Mt (266) 62 Sm 150.36 10 VIII 28 Ni 58.69 46 Pd 106.42 78 11 IB 94 95 Pu Am (244) (243) 13 IIIA 12 IIB 5 6 7 C 12.01 14 N 14.01 15 29 30 31 34 Si P 28.09 30.97 32 33 Cu Zn Ga Ge As Se 63.55 65.39 69.72 72.63 74.92 78.97 48 49 50 52 Ag Cd In Sn Sb Te 107.87 112.41 114.82 118.71 121.75 127.60 80 47 51 B 10.81 13 Al 26.98 14 15 IVA 16 VA VIA 79 81 82 Pt Au Hg TI Pb 195.08 196.97 200.59 204.38 207.2 110 111 112 113 114 Ds Rg Cn Nh Fl (271) (272) (277) (286) 83 Bi 208.98 115 16.00 16 52 S 32.07 84 Po (209) 17 VIIA 9 Md (258) F 19.00 17 Cl 35.45 35 Br 79.90 53 I 126.90 85 At (210) 116 Mc Lv (285) (289) (293) (294) Ts 117 66 70 63 64 65 67 68 69 Eu Gd Tb Dy Но Er Tm Yb 151.97 157.25 158.93 162.50 164.93 167.26 168.93 173.04 96 98 99 100 Cf Es Fm (251) (252) (257) 101 102 Cm (247) 97 Bk (247) 18. VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39.95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 No Lr (259) (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Explanation Metallic character is increased from top to bottom down the group because electron...View the full answer

Answered By

Raghuram Suru
I am a Mechanical Engineer, I am an honest and hard working person, I stick to my commitments, I am professional in my dealings. I am a good learner of new things, that makes me good in problem solving. My key strength includes leadership, creativity, troubleshooting skills and quick problem solving.
I have wide knowledge in mechanical engineering field and 10 years of experience. I am willing to solve mechanical engineering problems and work related to mechanical projects.I am expert in following subjects.
Machine Design
Engineering Mechanics
Mechanics of Machines
Manufacturing
Thermodynamics
Heat And Mass Transfer
Mechanical Vibrations
Heat And Mass Transfer
Refrigerant and Air conditioning
Fluid Mechanics
Engineering Mathematics
You will be definitely satisfied with the my work.If you want to contact me any time as I am available for you in 24 hours.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Which statement is true about trends in metallic character? a) Metallic character increases as you move to the right across a row in the periodic table and increases as you move down a column. b)...
-
State the trends in the periodic table for decreasing atomic radii. Periodic Table: 2 3 4 10 6 3 7 11 1 IA Li 6.94 Na 22.99 19 37 5 Rb K 39.10 4 87 2 IIA Fr (223) Be 9.01 12 Mg 24.31 20 38 Ca Sc...
-
Table 9.2 does not include francium because none of franciums isotopes are stable. Predict the values of the entries for Fr in Table 9.2. Predict the nature of the products of the reaction of Fr...
-
Use Figure 5.25 to encode or decode the messages in Problems 3748. Multiply by 4 and add 10. WDVCTWDVGLR JR VCT RZFNTWT KZP.TY.
-
Describe the institutional process for establishing generally accepted accounting principles for the federal government.
-
In January 2009, the company made $24,000 in expenditures. These expenditures should have been expensed immediately. Instead, the company recorded this $24,000 payment as a purchase of equipment with...
-
Locate the income statement of a company in each of the following industries: pharmaceutical, technology, retailergroceries, and automobile manufacturer. Calculate the gross profit margin, operating...
-
Selected comparative statement data for Navin Products Company are presented on the next page. All balance sheet data are as of December 31. InstructionsCompute the following ratios for 2013.(a)...
-
Why are risk-free spreads a valuable tool for assessing discount rates in a foreign market?
-
Predict the ionic charge for an aluminum ion, Al ?+ , based on the position of the element in the periodic table. Periodic Table: 2 3 4 15 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 Rb R 4 87 2 IIA Be...
-
Predict the ionic charge for a sodium ion, Na ?+ , based on the position of the element in the periodic table. Periodic Table: 2 3 4 5 6 7 Li 6.94 11 Na 22.99 19 1 IA K 39.10 37 55 4 2 IIA 87 Be 9.01...
-
In the system of accounting as used in the UK which of the following is not recorded in an account? (a) Loss of goods bought for sale (b) Goods removed by the owner for own use (c) Cash discount (d)...
-
A bank manager finds that about six customers enter the bank every 5 min. If the customer arrival time follows an exponential distribution, what is the probability that the next customer will arrive...
-
Suppose the average time a customer waits at the check-out line in a grocery store is 12 min with a standard deviation of 3 min. If you take a random sample of five customers, what is the probability...
-
The campus bus at Haverford College is scheduled to arrive at the business school at 8:00 a.m. Usually, the bus arrives at the bus stop during the interval 7:568:03. Assume that the arrival time...
-
A sample of 100 former basketball players from Slam Dunk University shows that 55 of the players graduated in 4 years. Construct a 90 % confidence interval for the proportion of basketball players...
-
As a CEO, you are trying to decide whether to acquire a foreign firm. The size of your firm will double after this acquisition to become the largest in your industry. On the one hand, you are excited...
-
The U.S. Department of Agriculture publishes data annually on various selected farm products. Shown here are the unit production figures (in millions of bushels) for three farm products for 10 years...
-
The registrar of a college with a population of N = 4,000 full-time students is asked by the president to conduct a survey to measure satisfaction with the quality of life on campus. The following...
-
On December 31, 2010, Shell hammer Co. sold 6-month old equipment at fair value and leased it back. There was a loss on the sale. Shell hammer pays all insurance, maintenance, and taxes on the...
-
The financial statements of P&G are presented in Appendix 5B or can be accessed at the books companion website, HYPERLINK "www.wiley.com/college/kieso" www.wiley.com/college/kieso. Refer to P&Gs...
-
Go to the books companion website or the company websites and use information found there to answer the following questions related to UAL, Inc. and Southwest Airlines. (a) What types of leases are...
-
Eleven-Eleven, Inc. creates guided meditation programs for individuals. On November 30, 20Y9, the balances of selected accounts of Eleven-Eleven, Inc. are as follows: Accounts Payable $67,800 Prepaid...
-
Create a discussion post to share with your classmates about a few tips or tricks that help you prepare for an interview.If you have not yet experienced an interview, feel free to discuss strategies...
-
Prepare horizontal analysis of the change in key accounts; at least 3 ratios Prepare vertical analysis of key expense accounts (amount as a percent of sales); at least 2 ratios for each year Falls...

Study smarter with the SolutionInn App


