Predict the ionic charge for a sodium ion, Na ?+ , based on the position of the
Question:
Predict the ionic charge for a sodium ion, Na?+, based on the position of the element in the periodic table.
Periodic Table:
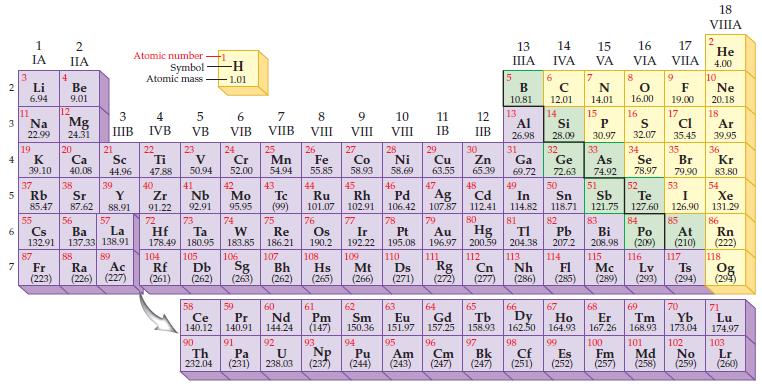
Transcribed Image Text:
2 3 4 5 6 7 Li 6.94 11 Na 22.99 19 1 IA K 39.10 37 55 4 2 IIA 87 Be 9.01 12 Mg 24.31 20 Ca 40.08 38 21 Rb Sr Y 85.47 87.62 88.91 88 Sc 44.96 3 4 IIIB IVB 39 56 Cs La Ba 132.91 137.33 138.91 57 89 Atomic number - Symbol Atomic mass Fr Ra Ac (223) (226) (227) 22 Ti 47.88 40 Zr 91.22 72 104 5 VB Rf (261) 23 V 50.94 41 Nb 92.91 Hf Ta 178.49 180.95 73 105 58 Ce 140.12 90 -H 1.01 Th 232.04 6 VIB 24 Cr 52.00 Db Sg (262) (263) 106 42 Tc Mo 95.95 (99) 74 59 Pr 140.91 7 VIIB 75 Re 183.85 186.21 91 25 Pa (231) Mn 54.94 43 107 Bh (262) 60 Nd 144.24 92 8 VIII 26 Fe 55.85 44 Ru 101.07 76 Os 190.2 61 9 VIII 93 27 U NP 238.03 (237) Co 58.93 62 Pm Sm (147) 150.36 10 VIII 45 46 Rh Pd 102.91 106.42 94 28 29 30 Ni Cu Zn 58.69 63.55 65.39 11 IB 63 Eu 151.97 95 Pu Am (244) (243) 47 5 13 IIIA 48 Cd 112.41 80 Hg 200.59 13 12 IIB B 10.81 7 N 14.01 15 Al Si P S 26.98 28.09 30.97 32.07 14 16 14 15 IVA VA 31 Ga 69.72 49 In 114.82 81 6 C 12.01 16 VIA 50 53 Ag 77 78 82 51 52 Sn Sb Te I 107.87 118.71 121.75 127.60 126.90 79 83 84 Pt Au Pb Bi Po 195.08 196.97 207.2 208.98 (209) 110 111 116 Hs Mt Ds Lv (265) (266) (271) (272) (277) (286) (285) (289) (293) (294) Ir 192.22 109 108 204.38 113 Nh 118 112 Cn Rg 114 FI 115 Mc Ts Og (294) 8 66 64 65 67 Gd Tb Dy Ho 157.25 158.93 162.50 164.93 96 97 Cm Bk (247) (247) O 16.00 32 33 34 Ge As Se 72.63 74.92 78.97 17 VIIA Br 79.90 85 18 VIIIA 9 10 F Ne 19.00 20.18 17 18 Cl Ar 35.45 39.95 35 36 At (210) 2 117 He 4.00 Kr 83.80 54 Xe 131.29 86 Rn (222) 68 70 71 174.97 69 Er Tm Yb Lu 167.26 168.93 173.04 98 99 100 101 102 103 Cf Es Fm Md No Lr (251) (252) (257) (258) (259) (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
We refer to the periodic table to find the group number of the element Sodium is in Group IA1 after ...View the full answer

Answered By

Rayan Gilbert
I have been teaching since I started my graduation 3 years ago. As a student, working as Teacher/PA has been tough but made me learn the needs for student and how to help them resolve their problems efficiently. I feel good to be able to help out students because I'm passionate about teaching. My motto for teaching is to convey the knowledge I have to students in a way that makes them understand it without breaking a sweat.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Predict the ionic charge for a phosphorus ion, P ? , based on the position of the element in the periodic table. Periodic Table: 2 3 4 15 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 Rb R 4 87 2 IIA Be 9.01...
-
Predict the ionic charge for a chlorine ion, Cl ?- , based on the position of the element in the periodic table. Periodic Table: 2 3 4 5 6 7 Li 6.94 11 1 IA Na 22.99 19 37 55 4 2 IIA 87 Be 9.01 12 K...
-
Predict the ionic charge for an aluminum ion, Al ?+ , based on the position of the element in the periodic table. Periodic Table: 2 3 4 15 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 Rb R 4 87 2 IIA Be...
-
Use a calculator to obtain solutions correct to the nearest hundredth in Problems 4954. 0.02x +0.831x + 0.0069 = 0
-
Identify the principals of the Joint Financial Management and Improvement Program (JFMIP) and identify in which branch of the federal government each operates. What role does each one play in the...
-
The following errors in the accounting records of the Chipp & Simon Partnership were discovered on January 10, 2011. The partners share net income and losses as follows: 65%, Chipp; 35%, Simon. 1....
-
Income statements are presented for the Elf Corporation for the years ending December 31, 2024, 2023, and 2022. Required: Write a one-paragraph analysis of Elf Corporations profit performance for the...
-
Forte Company begins operations on April 1. Information from job cost sheets shows the following. Job 12 was completed in April. Job 10 was completed in May. Jobs 11 and 13 were completed in June....
-
How do you plan to finance a short-term and a long-term project? Explain what success looks like in both situations.
-
State the trends in the periodic table for decreasing metallic character. Periodic Table: 2 3 4 10 6 3 7 11 1 IA Li 6.94 Na 22.99 19 37 5 Rb K 39.10 4 87 2 IIA Fr (223) Be 9.01 12 Mg 24.31 20 38 Ca...
-
Which of the following groups has a predictable ionic charge of three negative? (a) Group IIIA/13 (b) Group IIIB/3 (c) Group VA/15 (d) Group VB/5 (e) Group VIIIA/18.
-
Jane has started a gift basket company that specializes in regional products. Several corporations use her baskets for the holidays, to welcome new employees, and for their sales staff to use as...
-
Suppose a bowler takes a random sample of 15 games she has bowled and finds the sample mean to be 172. She knows that the standard deviation of her score is 8. Construct a 99 % confidence interval...
-
Wolves are the planets most widespread land-based large mammals. They used to be humans most direct competitors for meat. As a result, the Big Bad Wolf occupied a center stage in our psyche as a...
-
Suppose the weight of a football team is uniformly distributed with a minimum weight of 175 lb and a maximum weight of 285 lb. (a) Find the mean weight of the team. (b) Compute the standard deviation...
-
Some argue that engaging in a learning race is unethical. Others believe that a learning race is part and parcel of alliance relationships, especially those with competitors. What do you think?
-
The manager of a department store notices that the amount of time a customer must wait before being helped is distributed uniformly between 1 and 4 min. Find the mean and variance of the time a...
-
The U.S. Bureau of Labor Statistics produces consumer price indexes for several different categories. Shown here are the percentage changes in consumer price indexes over a period of 20 years for...
-
You've been asked to take over leadership of a group of paralegals that once had a reputation for being a tight-knit, supportive team, but you quickly figure out that this team is in danger of...
-
Presented in Illustration 21-31 are the financial statement disclosures from the 2007 annual report of Tasty Baking Company. Answer the following questions related to these disclosures. (a) What is...
-
As discussed in the chapter, U.S. GAAP accounting for leases allows companies to use off???balance-sheet financing for the purchase of operating assets. International accounting standards are similar...
-
Daniel Hardware Co. is considering alternative financing arrangements for equipment used in its warehouses. Besides purchasing the equipment outright, Daniel is also considering a lease. Accounting...
-
Forensic accounting is interesting to me because it demands an analytical approach beyond the numerical data to unveil any potential criminal activity. Presently, I am employed at a bank where I have...
-
A solution of NaCl(aq)NaCl(aq) is added slowly to a solution of lead nitrate, Pb(NO3)2(aq)Pb(NO3)2(aq), until no further precipitation occurs. The precipitate is collected by filtration, dried, and...
-
In module 3, you will provide information using Apple's financial statements. -When does Apple's fiscal year end? -What is Apple's Gross profit for the last three years? -What items make up Apple's...

Study smarter with the SolutionInn App


