State the trends in the periodic table for decreasing atomic radii. Periodic Table: 2 3 4 10
Question:
State the trends in the periodic table for decreasing atomic radii.
Periodic Table:
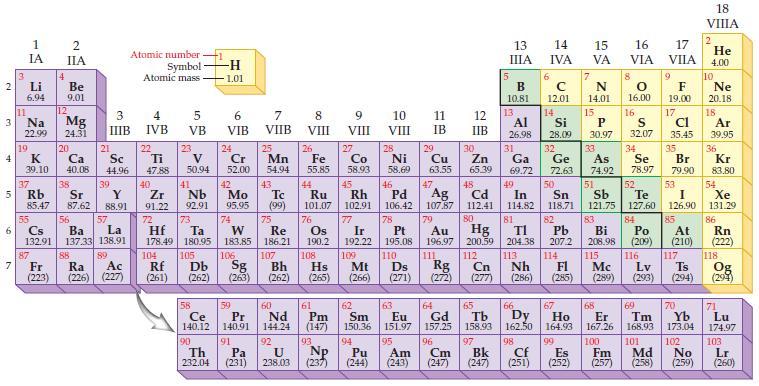
Transcribed Image Text:
2 3 4 10 6 3 7 11 1 IA Li 6.94 Na 22.99 19 37 5 Rb K 39.10 4 87 2 IIA Fr (223) Be 9.01 12 Mg 24.31 20 38 Ca Sc 40.08 44.96 21 Sr Y 85.47 87.62 88.91 55 56 La Cs Ba 132.91 137.33 138.91 88 3 4 IIIB IVB 39 57 89 Atomic number Symbol Atomic mass Ac Ra (226) (227) 22 Ti 47.88 5 VB 23 104 V 50.94 40 Nb Zr 91.22 92.91 41 105 -H 1.01 6 VIB 58 Ce 140.12 90 Th 232.04 24 Cr 52.00 42 Mo 95.95 74 72 73 W Re Hf Ta 178.49 180.95 183.85 186.21 106 Rf Db Sg (261) (262) (263) 59 Pr 140.91 91 7 VIIB Pa (231) 25 Mn 54.94 43 Tc (99) 75 107 60 Nd 144.24 8 VIII 92 26 27 Fe Co 55.85 58.93 Bh Hs (262) (265) 44 45 Ru Rh 101.07 102.91 76 Os 190.2 108 61 Pm (147) 9 VIII 93 U NP 238.03 (237) 77 Ir 192.22 109 Mt (266) 62 Sm 150.36 10 VIII 28 Ni 58.69 46 Pd 106.42 78 11 IB 94 95 Pu Am (244) (243) 13 IIIA 12 IIB 5 6 7 C 12.01 14 N 14.01 15 29 30 31 34 Si P 28.09 30.97 32 33 Cu Zn Ga Ge As Se 63.55 65.39 69.72 72.63 74.92 78.97 48 49 50 52 Ag Cd In Sn Sb Te 107.87 112.41 114.82 118.71 121.75 127.60 80 47 51 B 10.81 13 Al 26.98 14 15 IVA 16 VA VIA 79 81 82 Pt Au Hg TI Pb 195.08 196.97 200.59 204.38 207.2 110 111 112 113 114 Ds Rg Cn Nh Fl (271) (272) (277) (286) 83 Bi 208.98 115 16.00 16 52 S 32.07 84 Po (209) 17 VIIA 9 Md (258) F 19.00 17 Cl 35.45 35 Br 79.90 53 I 126.90 85 At (210) 116 Mc Lv (285) (289) (293) (294) Ts 117 66 70 63 64 65 67 68 69 Eu Gd Tb Dy Но Er Tm Yb 151.97 157.25 158.93 162.50 164.93 167.26 168.93 173.04 96 98 99 100 Cf Es Fm (251) (252) (257) 101 102 Cm (247) 97 Bk (247) 18. VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39.95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 No Lr (259) (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The trends in the periodic table for decreasing atomic radii are as follows Atomic radii decrease from left to right across a period This is because t...View the full answer

Answered By

Rohail Amjad
Experienced Finance Guru have a full grip on various sectors, i.e Media, Insurance, Automobile, Rice and other Financial Services.
Have also served in Business Development Department as a Data Anlayst
4.70+
32+ Reviews
83+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
State the trends in the periodic table for decreasing metallic character. Periodic Table: 2 3 4 10 6 3 7 11 1 IA Li 6.94 Na 22.99 19 37 5 Rb K 39.10 4 87 2 IIA Fr (223) Be 9.01 12 Mg 24.31 20 38 Ca...
-
Table 9.2 does not include francium because none of franciums isotopes are stable. Predict the values of the entries for Fr in Table 9.2. Predict the nature of the products of the reaction of Fr...
-
Researching the current availability of multicore processors and which processors can be teamed in multiprocessor systems. Provide examples and insightful observations, such as benefits, cost,...
-
Simplify the expressions in Problems 3138. (3x - 1) (x + 3x - 2)
-
The FASAB sets standards for federal agencies that relate to external financial reporting, much like the GASB sets standards for state and local governments for external financial reporting. Do you...
-
Refer to Practice 5-17. Compute the amount of cash received from the sale of the property, plant, and equipment.
-
The statement of cash flows segregates cash inflows and outflows by: (a) Operating and financing activities. (b) Financing and investing activities. (c) Operating and investing activities. (d)...
-
Ito Manufacturing Co. uses the process cost system. The following information for the month of December was obtained from the company's books and from the production reports submitted by the...
-
Find the value of each of these investments at the end of the period specified below. 2 (a) $3500 invested at 3-5% compounded annually for six years (b) $9000 invested at 8% compounded semiannually...
-
Predict the common ionic charge for Group IA/1 elements; Group IIA/2 elements; Group III/13 elements.
-
Predict the ionic charge for a chlorine ion, Cl ?- , based on the position of the element in the periodic table. Periodic Table: 2 3 4 5 6 7 Li 6.94 11 1 IA Na 22.99 19 37 55 4 2 IIA 87 Be 9.01 12 K...
-
You sold a security for $980 that you purchased five years before for $795. What was the holding period return? Prove that this return overstates the annualized, compound return.
-
The shelf life of hearing aid batteries is found to be approximated by an exponential distribution with a mean of 1/12 day. What fraction of the batteries would be expected to have a shelf life...
-
A study conducted in 1984 reported that the median pay in the United States was $18,700. What difficulties do you see in using this type of study for assessing incomes? Would you be willing to use...
-
A company has just installed a new automatic milling machine. The time it takes the machine to mill a particular part is recorded for a sample of 9 observations. The mean time is found to be X =...
-
The chief financial officer at Venture Corporation believes that an investment in a new project will have a cash flow in year one that is uniformly distributed between $1 million and $10 million....
-
An insurance company is interested in the average claim on its auto insurance policies. It believes the claims are normally distributed. Using the last 37 claims, it finds the mean claim to be $1,270...
-
Virginia Semiconductor is a leading manufacturer of prime silicon substrates. The company, situated in Fredericksburg, Virginia, was founded in 1978 by Dr. Thomas G. Digges and his brother, Robert....
-
A local politician is concerned that a program for the homeless in her city is discriminating against blacks and other minorities. The following data were taken from a random sample of black and...
-
On January 1, 2011, Irwin Animation sold a truck to Peete Finance for $33,000 and immediately leased it back. The truck was carried on Irwins books at $28,000. The term of the lease is 5 years, and...
-
Access the glossary (Master Glossary) to answer the following. (a) What is a bargain-purchase option? (b) What is the definition of incremental borrowing rate? (c) What is the definition of estimated...
-
What comprises a lessees minimum lease payments? What is excluded?
-
1) Does Newton Community have sufficient cash balances to cover the forecast risks in the initial three years before ASC operations reach full capacity? 2) What crucial assumption if altered, could...
-
Given the following information, calculate the effective gross income multiplier. Sale price: $950,000 Potential Gross Income: $250,000 Vacancy and Collection Losses: 10% Miscellaneous Income: $50,000
-
An asset used in a four-year project falls in the five-year MACRS class for tax purposes. The asset has an acquisition cost of $4,750,000 and will be sold for $1,375,000 at the end of the project. If...

Study smarter with the SolutionInn App


