Question: Figure P.11.44 depicts a single saw tooth function and its autocorrelation. Explain why cÆÆ (X) is symmetrical about the origin. Why does it extend from
Figure P.11.44
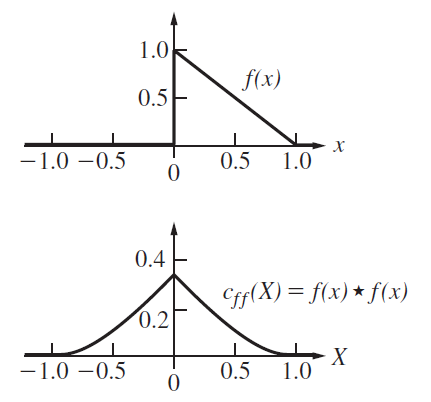
1.0 f(x) 0.5F 1.0 -1.0 0.5 0.5 0.4 Cff(X) = f(x) * f(x) 0.2 -1.0 0.5 0.5 1.0
Step by Step Solution
3.34 Rating (163 Votes )
There are 3 Steps involved in it
Symmetry is a fundamental property of autocorrelation The maxim... View full answer

Get step-by-step solutions from verified subject matter experts


