For each of the following, write a mechanism that accounts for the reaction shown. a) b) c)
Question:
For each of the following, write a mechanism that accounts for the reaction shown.
a)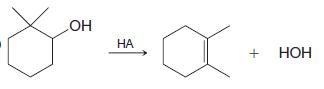
b)
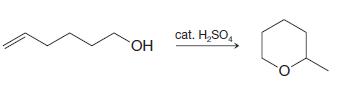
c)
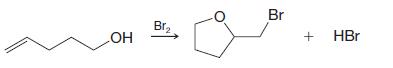
d)
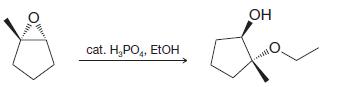
Transcribed Image Text:
OH HA + НОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
Answer a CH3CH2Br NaOH CH3CH2OH NaBr Mechanism 1 NaOH acts as a base and deprotonates the bromide io...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Write a mechanism that accounts for the following reaction: OH HA + HOH
-
For each of the following reaction coordinate diagrams, tell whether the structure of the transition state will more closely resemble the structure of the reactants or the structure of the products:...
-
Write structural formulas for each of the following alcohols and alkyl halides: (a) Cyclobutanol (e) 2,6-Dichloro-4-methyl-4-octanol (b) sec-Butyl alcohol (f) trans-4-tert-Butylcyclohexanol (c)...
-
For the curve defined by F(t) = (etcos(t), e* sin(t)) find the unit tangent vector, unit normal vector, normal acceleration, and tangential acceleration at 5A t 6 T N (5r) = 6 5T 6 aN ||
-
2(x + 3) 8 Describe the solution set as an inequality, in interval notation, and on a graph.
-
Walker Patel is a portfolio manager at an investment management firm. After successfully implementing meanvariance optimization (MVO), he wants to apply reverse optimization to his portfolio. For...
-
Use a stem-and-leaf plot to display the data shown in the table at the left, which represent the drunk driving cases registered at 30 strategic road intersections. Organize the data using the...
-
Pate Bros. uses the allowance method to account for uncollectible accounts expense. Pate experienced the following four events in 2013: 1. Recognized $68,000 of revenue on account. 2. Collected...
-
Please follow the link and read the canvas project. Based on the reading, please answer the following questions . Link for the Startup Analysis Canvas Project:...
-
Prove that alphabeta pruning takes time O(2 m/2 ) with optimal move ordering, where m is the maximum depth of the game tree.
-
The following reaction does not produce the product shown. (a) Predict the major product from the conditions shown above, and write a detailed mechanism for its formation. (b) What reaction...
-
Evaluate the relative merits of your syntheses on the basis of selectivity and efficiency. (Decide which ones could be argued to be the best syntheses and which might be poorer syntheses.)
-
Refer to the information in Problem 5-3B and assume the periodic inventory system is used. Required 1. Compute cost of goods available for sale and the number of units available for sale. 2. Compute...
-
The equation of motion of a spring-mass-damper system subjected to a harmonic force can be expressed as: * + 23 wx + w/2 = focoswt a) Find the steady-state response of the system in the form: x(t)= C...
-
The sign below is often seen on steep descents (Figure 1). It requires the driver to use the engine to retard the descent. Let's see why. A truck with a mass of 39 tonnes is travelling at the top of...
-
The sales for exercise equipment in a country were $1830 million in 1990 and $5102 million in 2005. (a) Use the regression feature of a graphing utility to find an exponential growth model and a...
-
a) State the law of variable proportions and highlights its key assumptions. (5 marks) b) Using an illustration, discuss the stages of production associated with the law of proportion and which will...
-
Stone Company is facing several decisions regarding investing and financing activities. Address each decision independently. 1. On June 30, 2024, the Stone Company purchased equipment from Paper...
-
Show that the only subspaces of R1 are {0} and R1 itself.
-
A random sample of 10 houses heated with natural gas in a particular area, is selected, and the amount of gas (in therms) used during the month of January is determined for each house. The resulting...
-
Explain which compound has a faster rate of SN1 reaction. a) c) HC CI CI or or J d) CI CHCI or or D CHCl OCH 3
-
Arrange these compounds in order of decreasing SN1 reaction rate. Ph CI CI CI Ph CI Ph
-
Show the products, including stereo chemistry, of these SN1 reactions: a) CHCH Ph CH3 + CHOH HC b) CHCH H CH, -CH-C-CI+ CHCOH CH
-
When owning and operating any business, whether it's a caf or a pool cleaning business, it's very important that you know exactly what your income and expenditure is. Is your business making money or...
-
Resources Unlimited Corporation was formed in 1985 through the merger of two natural pipeline companies. The result was the largest gas distribution network in the United States, with 38,000 miles of...
-
Toyota was the world's first automobile manufacturer to produce more than 10 million vehicles per year, which it has done since 2012, when it also reported the production of its 200-millionth...

Study smarter with the SolutionInn App


