Predict the product from each of the following reactions. (a) (b) (c) Br + B(OH) Pd catalyst
Question:
Predict the product from each of the following reactions.
(a)
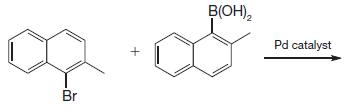
(b)
(c)
Transcribed Image Text:
Br + B(OH)₂ Pd catalyst
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
a The aryl bromide and the aryl borate react via SuzukiMiyaura re...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Convert the following into Chomsky Normal Form and Greibach Normal Form. 0 40 | 1B1 | BB C A S | A S|e C
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) OH SOC, pyr OH HBr NaNH2 OH OH (1) TsCI, pyr (2) EtSNa Nal, H2SO OH
-
Predict the organic product from each of the following oxidation reactions. (a) (b) (c) (d) (e) (1) KMnO4, HO. OH (2) H,O OH PCC CH2Cl2 OH (1) DMSO, (COCI)2 (2) EtgN OH H2CrO H2CrO
-
You are a financial analyst. Based on your analysis of Macys financial statements, provide a report to guide investors whether Macys is a buy (keep) or sell stock. Your report should include the...
-
After gaining enough experience to advance through a first salary schedule (A), police officers at the Los Angeles Police Department are paid annual salaries as described in Table 76 (Schedule 2). As...
-
Neutrinos are very low mass, extremely weakly interacting particles that permeate the universe. About a quadrillion will pass through you while you read this problem. There are multiple types, or...
-
Cold Appliances Pty Ltd makes refrigerators and is trying to determine the cost of its ending work in process. The accountant has put together the following data for the year ended 30 June 2025. Each...
-
ITown is a large computer discount store that sells computers and ancillary equipment and software in the town where State University is located. It has collected historical data on computer sales...
-
What do we know about the Galaxies over time according to https://webb.nasa.gov/content/science/galaxies.html What are some of the unknowns How might the JWT shed light on the galaxies over time?...
-
A 450-mm-long AISI 1020 steel rod is subjected to a tensile load of 55 kN. The allowable tensile stress is 140 MPa and the allowable total elongation is not to exceed 0.2 mm. Calculate the required...
-
The tranquilizing drug meprobamate (Equanil or Miltown) can be synthesized from 2-methylpentanal as follows. Give structures for meprobamate and for the intermediates AC: H HCHO, HO [A (C,H,O,)]...
-
Outlined here is a synthesis of 2-methyl-3-oxocyclopentanecarboxylic acid. Give the structure of each intermediate: Br OEt EtO OEt, EtO A (C2H2006) CN, EtO
-
1. What indicated that the terms in the agreement at issue in this case were accepted? 2. What were the appellant's arguments in support of her claim? Which of those contentions did the court imply...
-
A scuba diver and her gear displace a volume of 62.0 L and have a total mass of 65.8 kg. Part A What is the buoyant force on the diver in sea water? FB = Submit Request Answer Part B Will the diver...
-
Demand is strong and Nick Seamon can sell more sandwiches. In the short-run, how can his daily deli handle more volume? What will happen to the marginal product of labor at higher production? Why?...
-
Question 26 1 pts The mean volume for a bottle of cologne is 4 ounces and the standard deviation is 0.22 ounces. A random sample of 121 bottles is taken. What is the probability of obtaining a sample...
-
Mega Brothers Corporation has restaurants in almost 100 countries while Fast Snack Company is a much newer fast-food restaurant company, with restaurants throughout the United States and in several...
-
Explain why the court struck down the idea of 'separate but equal' ruling of Plessy v . Ferguson, and explain why you think that was an important event in American history.
-
Let U be a nonempty subset of a vector space V. Show that U is a subspace of V if and only if u1 + au2 lies in U for all u1 and u2 in U and all a in R.
-
If a force of F = 50 Ib is applied to the pads at A and C, determine the smallest dimension d required for equilibrium if the spring has an unstretched length of 1 ft. B 1 ft 1 ft F k = 15016/fr 1ft...
-
Explain which of the five products shown in is formed when 1-ethylcycpentene reacts with BH3 in THF, followed by treatment with NaOH andH2O2.
-
Explain which of the three products shown is formed when trans-2-butene reacts with CH2I2 andZn(Cu).
-
Explain which of the three products shown is formed when cis-2-butene reacts with OsO4 and t-BuOOH
-
Mrs. Sam Taffer was a brilliant engineer at IBM and made a lot of money from stock options. Since then, she has worked as a musician and raised her two sons. She wishes to have most of her property...
-
Miller Company ended its fiscal year on June 30, 2017. The company's adjusted trial balance as of the end of its fiscal year is shown below. MILLER COMPANY Adjusted Trial Balance June 30, 2017...
-
What depreciation method is used to write off property, plant and equipment? Regarding the triple bottom line, what information did the company disclose about environmental matters in the reports?...

Study smarter with the SolutionInn App


