Convert the following molecular model of aspirin into a line-bond structure, and identify the hybridization of each
Question:
Convert the following molecular model of aspirin into a line-bond structure, and identify the hybridization of each carbon atom (gray = C, red = O, ivory = H).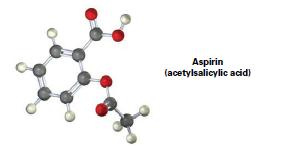
Transcribed Image Text:
Aspirin (acetylsalicylic acid)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
Aspirins chemical formula is C9H8O4 and its systematic name is acetylsalicylic ...View the full answer

Answered By

User l_1013947
I possess a comprehensive understanding of programming languages such as C++, Python, HTML, CSS, and Jupyter Notebook. These technical skills enable me to develop robust software solutions and create visually appealing web pages. With my expertise in coding, I can effectively tackle complex programming tasks and deliver high-quality results.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
In the hydrocarbon (a) What is the hybridization at each carbon atom in the molecule? (b) How many Ï bonds are there in the molecule? (c) How many Ï bonds? (d) Identify all the 120o bond...
-
Convert the following molecular model of ethane, C 2 H 6 , into a structure that uses wedged, normal, and dashed lines to represent three-dimensionality. Ethane
-
A chemical is spilled into a lake of pure water and the concentration of chemicals in this lake is 4 If 20 of the water in the lake is replaced with clean in one month then. What will be the...
-
A group of civic-minded merchants in Eldora organized the Committee of 100 for establishing the Community Sports Club, a not-for-profit sports organization for local youth. Each of the committees 100...
-
Explain the types of positive messages and the strategy used when business people write them.
-
SWIGART v. BRUNO CALIFORNIA COURT OF APPEALS 13 CAL. APP. 5TH 529 2017 According to the American Endurance Ride Conference, endurance riding is a highly competitive and demanding sport. It is...
-
The stockholders' equity section of the balance sheet for Brawner Company at December 31, 2012, is as follows: Required a. What is the par value per share of the preferred stock? b. What is the...
-
In a random walk suppose that at t = (n - 1)At the particle is located at x = max. The assumption is that at t = nAt the particle will have moved to x = (m+1)Ax with probability 3/4 or to x = (m-3)Ax...
-
Electrostatic potential maps of (a) acetamide and (b) methylamine are shown. Which of the two has the more basic nitrogen atom? Which of the two has the more acidic hydrogen atoms? (a) (b) Acetamide...
-
Is either of the following reactions likely to take place according to the pKa data in Table 1.2? (a) (b) HCN + CH3CO- Na+ Na+ CN + CH3COH
-
Describe what the project manager should do to perform the organizing function. Give some specific examples.
-
P5. (10 pts) Consider a slotted ALOHA system. Suppose there are 2N nodes sharing the channel. These nodes are divided in two groups with N nodes each. The probability that a node in group 1 and group...
-
Critique review of Love is Not a Pie attached Essay Instructions A critique is a formal evaluation of a text, and one of the most common forms of critique in literature is the review. In a review,...
-
For a water cooled ammonia condenser find the heat extracted if con- denser surface area 3 m, steel tube thickness 3 mm, condenser temperature 25C, average water temperature 18C, overall coeffi-...
-
A certain capacitor is made of two plates with a surface area of 2.4 m and separated by 7.2 m of an insulating material. If the capacitor is rated at 8 F, what is the material's dielectric constant?...
-
Describe how a rootkit works? What is adware, dumpster diving, backdoor, and botnets? What are the differences between Hacker and Cracker? What are the different types of hackers? Explain. How...
-
Refer to the information in the previous exercise. Find the mean and standard deviation of the total number of complaints received in a week. Having reached this point, you are concerned that the...
-
Express mass density in kg/m3 and weight density in lb/ft3. 1. Find the mass density of a chunk of rock of mass 215 g that displaces a volume of 75.0 cm3 of water. 2. A block of wood is 55.9 in. x...
-
a. How many different octapeptides can be made from the 20 naturally occurring amino acids? b. How many different proteins containing 100 amino acids can be made from the 20 naturally occurring amino...
-
Suppose you are trying to synthesize the dipeptide Val-Ser. Compare the product that would be obtained if the carboxyl group of N-protected valine were activated with thionyl chloride with the...
-
In determining the primary structure of insulin, what would lead you to conclude that it had more than one polypeptide chain?
-
The figure shows the displacement y of a traveling wave at a given position as a function of time and the displacement of the same wave at a given time as a function of position. Determine the...
-
Consider the circuit shown in the figure below. (Assume R b = + 25.0 V R R 20.0 R R a 15.0 and R2 = 3.50 2.) (a) Find the potential difference between points a and b. Your response differs from the...
-
Particle A has four times the speed and eight times the kinetic energy of particle B. What is the mass ratio m/mB

Study smarter with the SolutionInn App


