Convert the following molecular model of ethane, C 2 H 6 , into a structure that uses
Question:
Convert the following molecular model of ethane, C2H6, into a structure that uses wedged, normal, and dashed lines to represent three-dimensionality.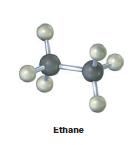
Transcribed Image Text:
Ethane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
Threedimensional geometry To represent the threedimensional geometry of a molecule we need to use a ...View the full answer

Answered By

User l_1013947
I possess a comprehensive understanding of programming languages such as C++, Python, HTML, CSS, and Jupyter Notebook. These technical skills enable me to develop robust software solutions and create visually appealing web pages. With my expertise in coding, I can effectively tackle complex programming tasks and deliver high-quality results.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Convert the following representation of ethane, C2H6. Into a conventional drawing that uses solid, wedged, and dashed lines to indicate tetrahedral geometry around each carbon (gray = c, ivory =H).
-
Convert the following molecular model of aspirin into a line-bond structure, and identify the hybridization of each carbon atom (gray = C, red = O, ivory = H). Aspirin (acetylsalicylic acid)
-
Assign R or S configuration to the chirality center in the following molecular model of the amino acid methionine (blue = N, yellow =S):
-
Why is an increase in price more likely to decrease the total revenue of a seller in the long run than in the short run?
-
Brookdale Hospital hired an inexperienced controller early in 20X4. Near the end of 20X4, the board of directors decided to conduct a major fund-raising campaign. They wished to have the December 31,...
-
How does graphic highlighting help save readers time?
-
Tom Bonacci brought his Jeep to Brewer Service Station to investigate a strange noise the vehicle was making. The Jeep was raised up on an automobile lift so that Brewer employee Paul Gebing could...
-
The city of Merlot operates a central garage through an Internal Service Fund to provide garage space and repairs for all city-owned and -operated vehicles. The Central Garage Fund was established by...
-
A space for storing boxes is 36 inches high. Each box is 6 inches high. A space of 9 inches must be left at the top.
-
Electrostatic potential maps of (a) acetamide and (b) methylamine are shown. Which of the two has the more basic nitrogen atom? Which of the two has the more acidic hydrogen atoms? (a) (b) Acetamide...
-
Is either of the following reactions likely to take place according to the pKa data in Table 1.2? (a) (b) HCN + CH3CO- Na+ Na+ CN + CH3COH
-
Will the bank statement show 'Dr' or 'Cr' if an account is overdrawn?
-
Related to the BMW Motorrad identify in which stage of the Product Life Cycle the company is and which tools of the IMC are more relevant in this stage. (minimum five hundred words )
-
D I AM ABLE TO RUNIT WITH OUT ERRORS> #include using namespace std; // Function to perform Warm-up exercise void performWarmUp(int warmUpReps) { cout < < "*** Warm-up Section ***" < < endl; for (int...
-
Briefly review the ten Principles outlined in Fr. Byron's book, "The Power of Principles" on Integrity, Veracity, Fairness (Justice), Human Dignity, Workplace Participation, Commitment, Social...
-
(a) Show that (2,1,6), (4,7,9) and (11,7,-12) are the vertices of a right triangle. (b) Find the area of the triangle.
-
Liam Co's December 31, 2021 and 2022 financial statements included the following information: 2022 2021 Accounts receivable $ 40,000 $ 36,000 Inventory 28,000 35,000 Net Sales 190,000 186,000 Cost of...
-
A restaurant manager receives occasional complaints about the quality of both the food and the service. The marginal probability distributions for the number of weekly complaints in each category are...
-
The first national bank pays a 4% interest rate compound continuously. The effective annual rate paid by the bank is __________. a. 4.16% b. 4.20% c. 4.08% d. 4.12%
-
Determine the amino acid sequence of a polypeptide from the following results: Acid hydrolysis gives Ala, Arg, His, 2 Lys, Leu, 2 Met, Pro, 2 Ser, Thr, Val. Carboxypeptidase A releases Val. Edmans...
-
How would a protein that resides in the interior of a membrane fold, compared with the water-soluble protein just discussed?
-
Which of the following parameters would be different for a reaction carried out in the presence of a catalyst, compared with the same reaction carried out in the absence of a catalyst? G , , , AS", ,...
-
A series circuit consists of a 20-2 resistor, a 25-mH inductor, and a 35-uF capacitor. If the frequency is 100 Hz, what is the power factor?
-
Consider a plane parallel-plate capacitor made of two strips of aluminum foil separated by a layer of paraffin-coated paper. Each strip of foil and paper is 3.80 cm wide. The foil is 0.00400 mm...
-
Two ropes are tightened in a hook as shown in the figure below. Determine the magnitude and direction of the resultant force. The two concurrent forces F, and F2 are 90 N and 290 N respectively. 45...

Study smarter with the SolutionInn App


