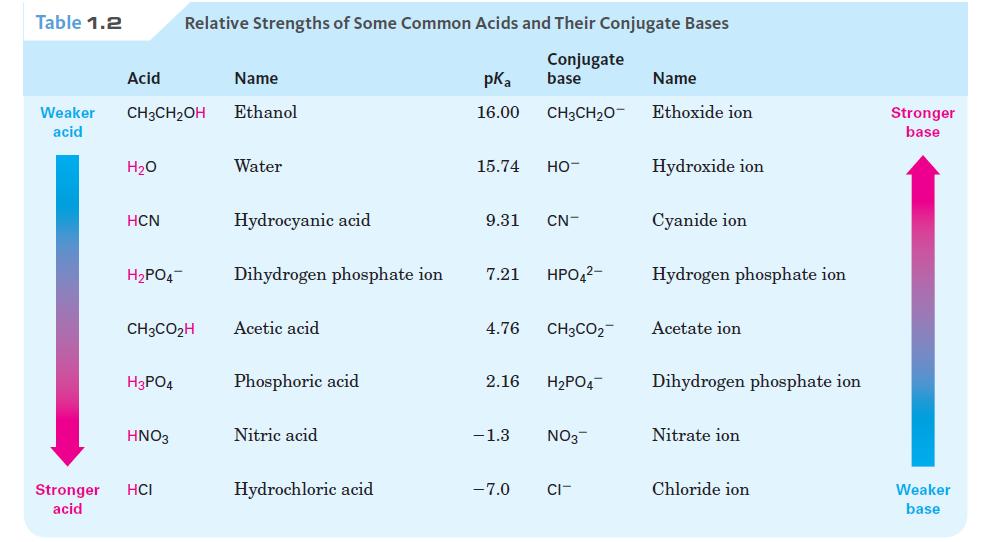
Is either of the following reactions likely to take place according to the pKa data in Table
Question:
Is either of the following reactions likely to take place according to the pKa data in Table 1.2?
(a)
![]()
(b)![]()
Transcribed Image Text:
HCN + CH3CO₂- Na+ Î Na+ CN + CH3CO₂H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
To determine whether the given reactions are likely to take place we need to compare the pKa values ...View the full answer

Answered By

Surendar Kumaradevan
I have worked with both teachers and students to offer specialized help with everything from grammar and vocabulary to challenging problem-solving in a range of academic disciplines. For each student's specific needs, I can offer explanations, examples, and practice tasks that will help them better understand complex ideas and develop their skills.
I employ a range of techniques and resources in my engaged, interesting tutoring sessions to keep students motivated and on task. I have the tools necessary to offer students the support and direction they require in order to achieve, whether they need assistance with their homework, test preparation, or simply want to hone their skills in a particular subject area.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Will either of the following reactions take place as written, according to the data inTable? (a) HCN + CH3CO2 Na+ Na* "CN CHCO2H Na* "CN CH3CH20- Na* |(b) CH3CH2OH HCN
-
The phenomenon of configuration inversion in a chemical reaction was discovered in 1896 by Paul Walden (Section 6.6). Walden's proof of configuration inversion was based on the following cycle: (a)...
-
summarize this article TITLE Overhead cost pools. By: Lambert III, S.J., Chen, Kung H., Internal Auditor, 00205745, Oct96, Vol. 53, Issue 5 DATA BASE Business Source Complete Internal auditors who...
-
Welch Company prepares monthly financial statements. Below are listed some selected accounts and their balances in the September 30 trial balance before any adjustments have been made for the month...
-
Friendly College is a small, privately supported liberal arts college. The college uses a fund structure; however, it prepares its financial statements in conformance with ASC 958. Partial balance...
-
When does persuasion become unethical and what is doublespeak?
-
Mrs. Palsgraf was waiting for a train on a platform of a railroad. When a different train came into the station, two men ran to get on that train before it left the station. While one of the men...
-
Essence of Esther Cosmetics Company is planning a one- month campaign for September to promote sales of one of its two cosmetics products. A total of $ 150,000 has been budgeted for advertising,...
-
1. Suppose that x1,. ,...,xk+1 is a circuit of length k in a graph G (x = xk+1) of length 3 or greater (k 3). Suppose as well that the circuit does not repeat edges (ie xx+1 xjxj+1 for i j). Show...
-
Convert the following molecular model of ethane, C 2 H 6 , into a structure that uses wedged, normal, and dashed lines to represent three-dimensionality. Ethane
-
Solve the given equation a log a M =
-
Go to the IRS Web site, www.irs.gov and, under "Learn About IRS," select "Tax Stats, Facts, and Figures." Then consult statistics regarding the gift tax. Prepare a brief write-up concerning the...
-
What are the inherent risks to fixed assets that the auditor must consider when planning the audit?
-
This was the first time in history that a cyberattack was synchronized with a military campaign. A. World War III B. Ukrainian Revolution C. Russo-Georgian War D. Civil War
-
What is a reporting requirement required under the Proceeds of Crime (Money Laundering) and Terrorist Financing Act ("PCMLTFA")?
-
The lengthy lockdowns caused by the Covid-19 pandemic have shown how precarious such oversight is in practice. Business interruption has caused trusted and approved suppliers to go bust, while an...
-
Christine is interested in purchasing a new printing press. She tests the output and calculates a Cpk of 0.73 whereas the current machine has a Cpk of 1. What should Christine do?
-
A college bookseller makes calls at the offices of professors and forms the impression that professors are more likely to be away from their offices on Friday than any other working day. A review of...
-
Listed below are several terms and phrases associated with basic assumptions, broad accounting principles, and constraints. Pair each item from List A (by letter) with the item from List B that is...
-
An alcohol will not react with aziridine unless the reaction is catalyzed by an acid. Why is a catalyst necessary? H HCl H;NCH,CH,OCH; CI CH;OH aziridine
-
The rate constant for the uncatalyzed reaction of two molecules of glycine ethyl ester to form glycylglycine ethyl ester is 0.6 s -1 M -1 . In the presence of [Co(ethylenediamine) 2 ] 2+ , the rate...
-
What causes the mode of catalysis to change from general base to nucleophilic in the hydrolysis of an ortho carboxyl-substituted phenyl acetate?
-
Part A Billiard ball A of mass m = 0.123 kg moving with speed VA = 2.80 m/s strikes ball B, initially at rest, of mass mB = 0.145 kg. As a result of the collision, ball A is deflected off at an angle...
-
Assume a mortgage loan has an outstanding principal balance of $100,000 and the interest rate is 6%. Calculate what portion of a $599.55 monthly payment will be allocated to interest.?
-
Find a website that you like and one that you do not like, which are respectful and abide by Saint Leo University ethics. For each website, identify who the target audience is, how it is organized...

Study smarter with the SolutionInn App


