For each pair of compounds, identify how you would distinguish them using either 1 H NMR spectroscopy
Question:
(a)
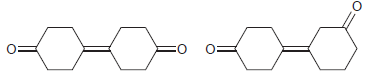
(b)
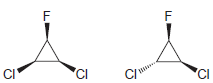
(c)
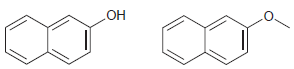
(d)
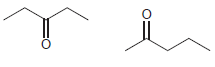
Transcribed Image Text:
'CI CI CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a The first compound will have four signals in its 13 C NMR spectrum while the second compound will ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each pair of compounds, circle the compound you expect to have the higher boiling point. Explain your reasoning. (a) (CH3)3C-C(CH3)3 and (CH3)2CH-CH2CH2-CH(CH3)2 (b) CH3(CH2)6CH3 and...
-
For each pair of compounds, describe which instrumental technique (IR, MS, proton NMR, carbon NMR) you could use to distinguish for certain which of the two compounds was in a sample. Describe what...
-
For each pair of compounds below, identify the more acidic compound: (a) (b) (c) (d) (e) (f) (g) (h) SH
-
What are the lessons of the 1997 Asian financial crisis? Were the IMF supported adjustment programs successful in their objective? Were there flaws in their design?
-
What are three major components of a social media information system?
-
Stan and Francine want to make perfume. In order to get the right balance of ingredients for their tastes, they bought 3 ounces of rose oil at \($2.04\) per ounce, 5 ounces of ginger essence for...
-
Graph the expectation function for the logistic growth model (12.34) for \(\theta_{1}=10, \theta_{2}=2\), and values of \(\theta_{3}=0.25,1,2,3\), respectively. Overlay these plots on the same set of...
-
A tool used for fabricating semiconductor devices consists of a chuck (thick metallic, cylindrical disk) onto which a very thin silicon wafer (p = 2700 kg/m 3 c = 875 J/kg K, k = 177 W/m K) is...
-
a) Figure la shows an overall two-port network consisting of Network A and Network B in a certain interconnection. Network A and Network B are respectively a T-network and a -network as shown....
-
An investor recently purchased a corporate bond that yields 9%. The investor is in the 36% combined federal and state tax bracket. What is the bonds after-tax yield?
-
Predict the multiplicity of each signal in the 1 H NMR spectrum of the following compound:
-
A compound with molecular formula C 8 H 18 exhibits a 1 H NMR spectrum with only one signal. How many signals would you expect in the 13 C NMR spectrum of this compound?
-
Write balanced equations for the combustion of ethane gas and liquid ethanol (to give gaseous products). (a) Calculate the enthalpy of combustion of each compound. Which has the more negative...
-
Use the following standard death rates to calculate standardized mortality ratios (SMRs) for Green City and Red City given that there were 4,050 total deaths in Green City and 30,025 total deaths in...
-
Sandra would like to organize LAB ( a legal corporation ) as either an S corporation or a C corporation for tax purposes. In either form, the entity is expected to generate an 8 percent annual before...
-
The Alpine House, Incorporated, is a large retailer of snow skis. The company assembled the information shown below for the quarter ended March 3 1 : Amount Sales $ 1 , 4 5 2 , 0 0 0 Selling price...
-
Lee, Inc. acquired 3 0 % of Polk Corp. ' s voting stock on January 1 , Year 1 , for $ 1 0 0 , 0 0 0 . During Year 1 , Polk earned $ 4 0 , 0 0 0 and paid dividends of $ 2 5 , 0 0 0 . Lee's 3 0 %...
-
Thad, a single taxpayer, has taxable income before the QBI deduction of $ 2 1 2 , 1 0 0 . Thad, a CPA, operates an accounting practice as a single member LLC ( which he reports as a sole...
-
The following information was drawn from the records of Tristan Company: Accounts payable (ending) ......................................................$12,000 Cash collected from accounts...
-
Briefly describe the following types of group life insurance plans: a. Group term life insurance b. Group accidental death and dismemberment insurance (AD&D) c. Group universal life insurance d....
-
How could you distinguish between the isomers Cyclopentanecarboxylic acid and 4-hydroxycyclohexanone by 1H and 13C NMR spectroscopy?
-
Give IUPAC name for the following carboxylic acids (reddish brown = Br). (a) (b) (d) (c)
-
Would you expect the following carboxylic acids to be more acidic or less acidic than benzoic acid? Explain. (Reddish brown = Br) (a) (b) (d) (c)
-
What interventions are the most beneficial to the clients your agency serves? Explain
-
Using the following predicates: square (x) is true if x is a square (otherwise it is false) star (x) is true if x is a star (otherwise it is false) circ(x) is true if x is a circle (otherwise it is...
-
Comparative financial statements for Weller Corporation, a merchandising company, for the year ending December 31 appear below. The company did not issue any common stock during the year. A total of...

Study smarter with the SolutionInn App


