Compound A is a by-product of the autoxidation of cumene, and compound B is a by-product of
Question:
Compound A is a by-product of the autoxidation of cumene, and compound B is a by-product of the acid-catalyzed conversion of cumene hydroperoxide to phenol and acetone.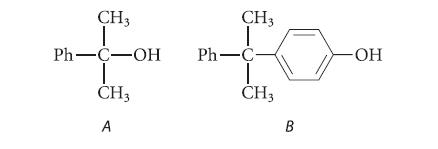
Draw a curved-arrow mechanism that shows how compound A can react with phenol under the conditions of Eq. 18.102 to give compound B.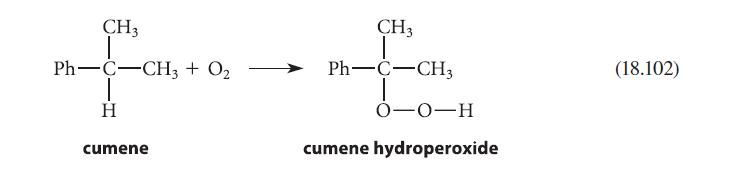
Transcribed Image Text:
CH3 Ph-C-OH T CH3 A CH3 Ph-C CH3 B -ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The alcohol A can form a relatively stable tertiary benzylic ca...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write a stepwise mechanism for the hydrolysis of chlorobenzene under the conditions shown in Table 24.3. TABLE 24.3 Industrial Syntheses of Phenol Reaction and comments Chemical equation Reaction of...
-
Draw the structure of each of the following. (Some parts may have more than one correct answer.) (a) A nine-carbon ether that can be prepared by the Williamson synthesis. (b) An ether that would...
-
In the conversion shown in Fig. P18.77, the Diels-Alder reaction is used to trap a very interesting intermediate by its reaction with anthracene. From the structure of the product, deduce the...
-
Modify the test client in Turtle to take an odd integer \(n\) as a command-line argument and draw a star with \(\mathrm{n}\) points.
-
Herb and Randy are twin jazz musicians who perform as a trombonesaxophone duo. At the age of twenty, however, Randy got an irresistible offer to join a road trip to perform on a star 15 light-years...
-
Determine the equal, annual, end-of-year payment required each year over the life of the loans shown in the following table to repay them fully during the stated term of theloan. Loan Principal...
-
Build an experimental setup that lets you monitor messages on an embedded network.
-
Mercury Delivery Service completed the following transactions and events involving the purchase and operation of equipment for its business. 2012 Jan. 1 Paid $ 25,860 cash plus $ 1,810 in sales tax...
-
In 2023, USCO, which is subject to a 21% U.S. corporate tax rate, has $1 Million of worldwide active income which is all from foreign sources in Country X. Assume USCO's foreign-source active income...
-
Give the product(s) (if any) expected when p-iodotoluene or other compound indicated is subjected to each of the following conditions. (a) CHOH, 25 C (b) CH3O- in CHOH, 25 C (c) CHO, pressure, heat...
-
(a) Give the structure of the product formed in the reaction of urushiol with K 2 CO 3 and a large excess of methyl iodide. (b) Would this compound be likely to provoke the same allergic skin...
-
Table 3-1(14-1) in the text shows the percentage undervaluation or overvaluation in the Big Mac, based on exchange rates in July 2019. Go to the main data repository for the BigMac dataset at...
-
The following table shows the investments of SP Ltd, which are classified into three sub-categories. Assumptions: (a) Returns are independently and normally distributed. (b) Each category is treated...
-
Consider the following cash flows on two mutually exclusive projects for Tomatina Recreation SA. Both projects require an annual return of 15 per cent. (a) If your decision rule is to accept the...
-
Explain the rationale for the accounting treatment of compound financial instruments under IAS 32. Do you agree with IAS 32s stance?
-
During 2007, the U.S. economy was hit by a price shock when the price of oil increased from around $60 per barrel to around $130 per barrel by June 2008. While inflation increased during the fall of...
-
Scenario A A Co acquired a controlling interest in B Co and entered into the following transactions on acquisition date, 1 July 20x3. Required: Prepare the journal entries that A Co would record to...
-
Study the Excel output. Explain the distribution in terms of shape and mean. Are these probabilities what you would expect? Why or why not? x Values Poisson Probabilities: = 2.78 0...
-
Read Case Study Google: Dont Be Evil Unless and answer the following: Why do you think Google was adamant about not wanting to supply information requested by the government concerning the Child...
-
1. Draw the important resonance forms for each compound. 2. Label the hybridization and bond angles around each atom other than hydrogen. 3. Use a three-dimensional drawing to show where the...
-
List each set of compounds in order of increasing boiling point. (a) Hexane, octane, and decane (b) Octane, (CH3)3 C-C(CH3)3 and CH3CH2C(CH3)2CH2CH2CH3
-
Draw a graph, similar to Figure 3-11, of the torsional energy of 2-methylbutane as it rotates about the C2¬C3 bond. Figure 3-11 3.8 kJ (0.9 kcal) 15 kJ 21 kJ (3.6 kcal) (5 kcal) -21 kJ (5 kcal)...
-
Pursuant to a complete liquidation, EB Corporation distributes the following assets to its unrelated shareholders: land held for three years as an investment ( basis of $ 3 0 0 K , fair market value...
-
After reading the fine print in your credit card agreement, you find that the "low" interest rate is actually an 17.05% APR, or 1.4208% per month. What is the effective annual rate?
-
In what ways can the resource-based view of the firm be used to analyze organizational capabilities and behavior?

Study smarter with the SolutionInn App


