Each of the following compounds protonates on nitrogen. Draw the conjugate acid of each. Which compound is
Question:
Each of the following compounds protonates on nitrogen. Draw the conjugate acid of each. Which compound is more basic? Explain.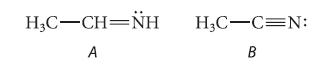
Transcribed Image Text:
HỌC–CH=NH H₂C-C=N: A B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The conjugate acids are as follows HCCHNH AH HCCNH ...View the full answer

Answered By

Pranav Makode
I am a bachelor students studying at professor ram meghe institute of technology and research. I have a great experience of being an expert. I have worked as an expert at helloexperts and solvelancer as a part time job. I have also worked as a doubt solver at ICAD SCHOOL OF LEARNING, which is in Amravati city. I have also worked as an Freelancer.
I have great experience of helping students, as described above. I can help any students in a most simple and understandable way. I will not give you have any chance for complaint. You will be greatfull to accept me as an expert.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Job costing is: A) used by businesses to price unique products for different jobs B) used to calculate the percentage of work completed C) used to calculate equivalent units D) used by businesses to...
-
Which of the following compounds are chiral? Draw each compound in its most symmetric conformation, star (*) any asymmetric carbon atoms, and draw any mirror planes. Label any meso compounds. You may...
-
Draw a structure for each of the following compounds in its more stable chair conformation. Explain your choice. (a) (b) CH3 CH3 CH (CH),C CHA CH, ," CH3 CH3
-
Mr. and Mrs. Thomas are buying a new house for $330,000. Compare the two loan offers by finding the monthly payments for each and calculating the total amount paid for each. Loan Options Down Payment...
-
The following data apply to Owens Sports, Inc., for April 2010: 1. Balance per the bank on April 30, $12,250. 2. Deposits in transit not recorded by the bank, $2,700. 3. Bank error; check written by...
-
The owners equity accounts for Hexagon International are shown here: Common stock ($1 par value) ....... $ 30,000 Capital surplus .............. 185,000 Retained earnings ............ 627,500 Total...
-
Jason Cannon purchased a defective tractor for which Cannon brought numerous causes of action against Bodensteiner Implement Company (Bodensteiner). One of the claims was breach of express...
-
The balance sheet of Phototec, Inc., a distributor of photographic supplies, as of May 31 is given below: Phototec, Inc., has not budgeted previously, and for this reason it is limiting its master...
-
The figure depicts Jack - in - the - box: "Jack" is attached inside a box by a spring, as shown. You estimate Jack's mass to be 0 . 4 k g . As so often happens, a sign tells you the spring constant:...
-
(a) Ion A is more acidic than ion B in the gas phase. Is this the acidity order predicted by hybridization arguments? Explain. (b) Ion B is less acidic because it is stabilized by resonance, whereas...
-
Give the product that results from the addition of one equivalent of Br 2 to 3-hexyne. What are the possible stereoisomers that could be formed?
-
Refer to the Journal of Deaf Studies and Deaf Education (Fall 2006) paired difference study on vocalized laughter among deaf users of sign language, presented in Exercise. Suppose you want to...
-
Why do investors and managers need to understand how to estimate a firms intrinsic value?
-
What is the difference between a stocks price and its intrinsic value?
-
What are some reasons a company might use classified stock?
-
A W. Hope's year ended on 30 June 2020. Write up the ledger accounts, showing the transfers to the financial statements and the balances carried down to the next year for the following: (a)...
-
What is the preemptive right, and what are the two primary reasons for its existence? Identify some actions that companies have taken to make takeovers more difficult.
-
Discuss the difference in the tax treatment of payments received from an employer-provided health and accident insurance policy and a health and accident insurance policy purchased by the taxpayer.
-
Answer the following two independent questions. a. MM Corporation is considering several proposed investments for the coming budget year. MM produces electrical apparatus for industrial complexes....
-
Benzene has an ultraviolet absorption at ? max = 204mm, and para-toluidine has ? max = 235nm. How do you account for this difference? -NH2 p-Toluidine U max = 235 nm) Benzene Amax = 204 nm)
-
What product would you expect to obtain from a nucleophilic substitution reaction of (S)-2-bromohexane with acetate ion, CH3CO2 ? Assume that inversion of configuration occurs, and show the stereo...
-
What product would you expect to obtain from SN2 reaction of OH with (R)-2-bromo-butane? Show the stereo chemistry of both reactant and product.
-
5. Counter design: (a) [6%] What is the difference between an asynchronous counter and a synchronous counter? (b) [8%] Design an asynchronous 3-bit counter using J-K flip-flops. Draw the figure. (c)...
-
6. [10%] The following figure shows a decade counter. Explain where and why the glitch happens in the timing. HIGH- 20 Q: Jo J 5 CLK C C C Ko K K K
-
Explain where on the MHC molecule CD4 and CD8 bind during interactions between T-cell receptors and MHC molecules.

Study smarter with the SolutionInn App


