Explain why the hydration of ethylene (Eq 4.44) is a very slow reaction. (Think about the structure
Question:
Explain why the hydration of ethylene (Eq 4.44) is a very slow reaction. (Think about the structure of the reactive intermediate and apply Hammond’s postulate.)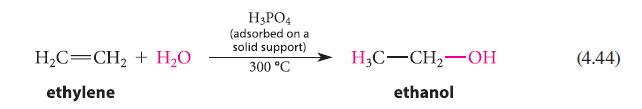
Transcribed Image Text:
H₂C=CH₂ + H₂O ethylene H3PO4 (adsorbed on a solid support) 300 °C H3C-CH₂-OH ethanol (4.44)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The intermediate in the hydration of ethylene is a primary carbocation Hence the transition s...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Explain why the hydration of this alkene occurs 1015 times faster than the hydration ofethene: OH H20 CH,CH,OCHCH3 CH.CH,OCH=CH, H,SO,
-
The rates of many atmospheric reactions are accelerated by the absorption of light by one of the reactants. For example, consider the reaction between methane and chlorine to produce methyl chloride...
-
KYC's stock price can go up by 15 percent every year, or down by 10 percent. Both outcomes are equally likely. The risk free rate is 5 percent, and the current stock price of KYC is 100. (a) Price a...
-
Administrative agencies, boards or commissions usually use registration or licensing to control business subject to the administrative law. True False
-
A new truck, manufactured by General Motors Corp. (GMC), stalled in rush hour traffic on a busy interstate highway because of a defective alternator, which caused a complete failure of the trucks...
-
Is kaizen budgeting the Japanese term for activity-based budgeting (ABB)?
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. Determine the IRR(s) for this project. b. Is this project economically attractive? EOY 0 1 2 3 4 5 6 NCF -$101 $411...
-
Office Automation, Inc., developed a proposal for introducing a new computerized office system that will improve word processing and interoffice communications for a particular company. Contained in...
-
11. Sinking Fund A company borrows $5000 on which it will pay interest at the end of each year at the annual rate of 11%. In addition, a sinking fund is set up so that the $5000 can be repaid at the...
-
Give the structures and IUPAC substitutive names of the isomeric alkenes with molecular formula C 6 H 12 containing five carbons in their principal chains.
-
Give the mechanism for the reaction in Eq. 4.43. Show each step of the mechanism separately with careful use of the curvedarrow notation. Explain why the rearrangement takes place. H HC-C-CH=CH + HO...
-
In Exercises explore the Alternating Series Remainder. (a) Use a graphing utility to find the indicated partial sum S n and complete the table. (b) Use a graphing utility to graph the first 10 terms...
-
If KBB Pizzeria has average cash and cash equivalents of $100,000, average current liabilities of $150,000, and average total liabilities of $200,000, what is KBB's quick ratio? Show your...
-
what is the difference between joinder of claims and joinder of parties?
-
What are your thoughts about "Anypersonsubject to military law for any crimeoroffensesmadepunishablebyArticles of War and any person who by law of war is subject to trial by military tribunals"? 2 ....
-
What is SAP system 2) Why SAP system is important in accounting? 3) Elaborate the problem statement with proper justification 4) Elaborate proposed solution should match with the problem statement...
-
QUESTION 1 The followings trial balance are extracted from WIKI Bhd. as at 31 December 2020. Land and building 200,000 Motor vehicles Machinery 90,000 80,000 8% Bond payable 100,000 Ordinary capital...
-
What is the most important source of revenue and the major type of expenditure at the Federal level? At the state level? At the local level?
-
1. Which of the four major types of information systems do you think is the most valuable to an organization? 2. How do you critically associate the ideas of business agility and business efficiency...
-
2, 3-Dimethyl-2, 3-butanediol has the common name pinacol. On heating with aqueous add, pinacol rearranges to pinacolone, 3, 3-dimethyl-2-hutanone. Suggest a mechanism for this reaction. CH :-C CH ...
-
As a rule, axial alcohols oxidize somewhat faster than equatorial alcohols. Which would you expect to oxidize faster, cis-4-tert-butylcyclohexanol or trans-4-tert-butylcyclohexanol? Draw the more...
-
Propose a synthesis of bicyclohexylidene, starting from Cyclohexanone as the only source of carbon. Bicyclohexylidene
-
What is a phonemic inventory? What is a phonetic inventory? How would someone determine the phonemic or phonetic inventory of a child (or adult)? What steps would you need to take to be able to...
-
A taxpayer files their return and decides to treat it with what is later to be determined as negligence in their failing to accurately report their total income. What is the amount of the penalty...
-
Carole owns 100% of Carole Inc, and her spouse Carl owns 100% of Carl Inc (both CCPCs). Carole is considering purchasing 30% of the common shares of Carl Inc. Explain to Carole how this transaction...

Study smarter with the SolutionInn App


