From what epoxide and what nucleophile could each of the following compounds be prepared? (Assume each is
Question:
From what epoxide and what nucleophile could each of the following compounds be prepared? (Assume each is racemic.)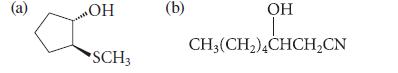
Transcribed Image Text:
(a) ОН SCH3 (b) OH CH3(CH₂)4CHCH₂CN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The strategy in this problem is to let the OH group originate from the epo...View the full answer

Answered By

Lisper Wanja
I am an experienced and highly motivated writer with a passion for the skills listed. I have a proven track record of my expertise and my aim is to deliver quality, well-detailed and plagiarism free projects. My genuine passion for writing combined with my ongoing professional development through school and research makes me an ideal candidate within for any assignment.
4.90+
233+ Reviews
388+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
a. How could each of the following compounds be prepared from a hydrocarbon in a single step? 1. 2. 3. b. What other organic compound would be obtained from each synthesis? OH Br Br CH CH HO
-
From what epoxide and what nucleophile colld each of the following compound be prepared Inppued? (Assums each is racemic.) C,H OH/H,O CH CH2 sodium azide
-
From which alkyne could each of the following compounds be prepared by acid- catalyzed hydration? (a) (c) CH3CCHCHCH3 (b) O (CH3)3C-C-CH3
-
1. Describe briefly how computers work and become tools for committing cyber-related crimes. 2. Use of proxies and VPNs is one of the methods cybercriminals try to cover their digital track and...
-
John Verner is the controller for BioMedic, Inc., a biotechnology company. John is finishing his preparation of the preliminary financial statements for a meeting of the board of directors scheduled...
-
Key comparative figures ($ millions) for both NIKE and Reebok follow: Required: a. Compute common-size percents for both companies using the data provided. b. Which company incurs a higher percent of...
-
Conventional financial accounting suggests that environmental permits should be measured by using the historical cost principle. One alternative is that current market prices should be used instead....
-
Hart Corporation is a chemical company that produces cleaning fluids of different types; it is the main employer in a small town. Stan Hart has been the company president for 15 years and is paid a...
-
Alex, Carol, and Ted are owners of a closely held corporation and have entered into an entity plan buy - sell agreement known as a stock redemption agreement. How many life insurance policies are...
-
Explain each of the following facts with a mechanistic argument. (a) When butyl methyl ether (1-methoxybutane) is treated with HI and heat, the initially formed products are mainly methyl iodide and...
-
The chlorohydrin trans-2-chlorocyclohexanol reacts rapidly in base to form an epoxide. The cis stereoisomer, however, is relatively unreactive and does not give an epoxide. Explain why the two...
-
Assume that Santana Rey expands Business Solutionss system to include special journals. Required 1. Locate the transactions related to January through March 2022 for Business Solutions in Chapter 5....
-
Could a contract be difficult to categorize as either public or private international law? What would such a contract look like?
-
As you have seen in this chapter, international trade law is founded on the importance and benefits of removing trade barriers. However, WTO rules also recognize the need to restrain trade in certain...
-
Calculate the mean, variance, and skewness coefficient for the data given in Table 4.9. Is the distribution symmetric ? Table 4.9 Year 90 91 92 93 94 95 96 97 98 99 00 01 02 03 04 05 06 07 08 09 Sum...
-
The United States brought a lawsuit requesting forfeiture of a \($38.5\) million jet purchased by Teodoro Nguema Obiang Mangue (Nguema) because the government believed the jet had been purchased with...
-
In 1990 Congress passed the Gun-Free School Zone Act that made it unlawful to possess a firearm in a school zone. A highschool senior carried a concealed and loaded handgun into his high school and...
-
A housekeeping support department budgets its costs at $40,000 per month plus $12 per hour. For November the following were the estimated and actual hours provided by the housekeeping support...
-
The ultimate goal of Google, Bing, and other consumer search engines is to provide users with search listings that contain useful information on the topic of their search. What recommendations would...
-
Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material for the biosynthesis of mevalonate, as shown in figure. At what positions in mevalonate...
-
Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material and that the mevalonate pathway is followed. Identify the positions in ?-cadinol where...
-
Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material and that the mevalonate pathway is followed. Identify the positions in squalene where...
-
Examine one recent health-related judicial decision, or a health-related topic that is currently being litigated for the following:...
-
Reading the information provided, please provide a detailed response to the material. Include at least one key point in-text citation and two references to support your response no older tfivean five...
-
TJ runs a side business called TJ's Tikoy Shop that becomes busy every year around the New Year selling tikoy (rice cakes/). To prepare for the rush, he hires extra staff to help fill the order. Each...

Study smarter with the SolutionInn App


