Give the structure of the organic product expected when CH 2 I 2 reacts with each of
Question:
Give the structure of the organic product expected when CH2I2 reacts with each of the following alkenes in the presence of a Zn–Cu couple: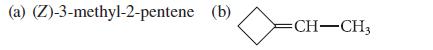
Transcribed Image Text:
(a) (Z)-3-methyl-2-pentene (b) CH-CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a b HC H CHCH3 CH3 ...View the full answer

Answered By

Anjali Arora
Having the experience of 16 years in providing the best solutions with a proven track record of technical contribution and appreciated for leadership in enhancing team productivity, deliverable quality, and customer satisfaction. Expertise in providing the solution in Computer Science, Management, Accounting, English, Statistics, and Maths.
Also, do website designing and Programming.
Having 7 yrs of Project Management experience.
100% satisfactory answers.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the structure of the expected product formed when benzylamine reacts with each of the following reagents: (a) Hydrogen bromide (b) Sulfuric acid (c) Acetic acid (d) Acetyl chloride (e) Acetic...
-
Give the structure of the expected product formed when benzylamine reacts with each of the following reagents: (a) Hydrogen bromide (b) Sulfuric acid (c) Acetic acid (d) Acetyl chloride (e) Acetic...
-
Give the structure of the product formed when each of the following alkenes reacts with bromine in water: (a) 2-Methyl-1-butene (c) 3-Methyl-1-butene (b) 2-Methyl-2-butene (d) 1-Methylcyclopentene
-
The Acme Insurance Company purchased a five-year bond whose interest rate floats with LIBOR. Specifically, the interest rate in a given year is equal to LIBOR plus 200 basis points. At the same time...
-
Assume at the beginning of 2012, the Ashlawn Village Street and Highway Fund (a special revenue fund) has cash of $300,000 offset by assigned fund balance in the same amount. 1. During the year, the...
-
A memorandum by Labor Secretary Robert Reich to President Bill Clinton suggested that the government penalize U.S. companies that invest overseas rather than at home. According to Reich, this kind of...
-
Reconsider the data from Problem 56. What is the capital recovery cost of Model 334A? Data from problem 56 Octavia Bakery is planning to purchase one of two ovens. The expected cash flows for each...
-
Martinez Companys ledger shows the following balances on December 31, 2010. 5% preferred stock$10 par value, outstanding 20,000 shares $ 200,000 Common stock$100 par value, outstanding 30,000 shares...
-
Use the below table to answer the following questions. Selling Price $43.00 = Sales Volume Variable 2,200 3,200 Fixed Cost Cost 4,200 Profitability 5,200 6,200 $47,200 15 $14,400 $42,400 $70,400...
-
Predict the products that result when each of the following alkenes reacts with chloroform and potassium tert-butoxide. Give the structures of all product stereoisomers, and, if more than one...
-
(a) Give the structures of two isomeric alkylmagnesium bromides that would react with water to give propane. (b) What compounds would be formed from the reactions of the reagents in (a) with D 2 O?
-
Determine whether the following variables are qualitative or quantitative. (a) Gender (b) Temperature (c) Number of days during the past week that a college student studied (d) Zip code Approach...
-
Examine the range of theories underpinning job design, noting any shortcomings.
-
Explain what is meant by: (a) attribution theory; and (b) impression management.
-
Highlight signifcant points in the debate about the impact of new technology on job design, in particular recent developments in AI and automation.
-
Why is reinforcement theory a learning theory considered in the context of motivation?
-
After consultation with its employees, Sunshine Company decides to switch the employee pay period to biweekly instead of weekly. Sunshines weekly payroll is $925,000. The annual cost of funds is 12%....
-
Gene, Inc., invested in a machine with a useful life of six years and no salvage value . It depreciated the machine using the straight-line method; the machine was expected to produce a $20,000...
-
1. Using the information from Problem 16-4B, prepare a statement of cash flows for Lim Garden Supplies Inc. using the direct method of presenting cash flows from operating activities. 2. How does Lim...
-
Hydrocarbon A has the formula C 9 H 12 and absorbs 3 equivalents of H 2 to yield R, C 9 H 18 , when hydrogenated over a Pd/C catalyst. On treatment of A with aqueous H 2 SO 4 in the presence of...
-
How would you carry out the followingreactions? (a) CCH-CCH3 CH3CH2C=CH () H2C%3CH CH3CH2CH2CHO .3- (c) 3C (d) CH . CH 7, 22H (e) CH3CH2C=CH (f) CH3CH2CH2CH2CH=CH2 CHCH2CH2CH2C%CH (2 steps)
-
Occasionally, chemists need to invert the stereochemistry of an alkene?that is, to convert a cis alkene to a trans alkene, or vice versa. There is no one-step method for doing an alkene inversion,...
-
When choosing business entities, you need to take many factors into account. For this paper, you will consider what you have learned about S corporations. Think about a company's tax plan and how...
-
When a purchaser requests the sales deposit to be invested, what special purpose Trust Account will the amount be deposited in?
-
David and Sherrill divorced effective in 2020. Last year Sherrill paid her ex-husband David $50,000. Per the divorce decree $30,000 of this was child support and $20,000 was alimony. How much of the...

Study smarter with the SolutionInn App


