As mentioned in problem 5.58, some molecules are chiral even though they lack a chirality center. For
Question:
a.
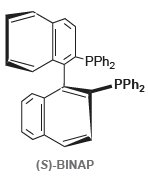
b.
Transcribed Image Text:
PPH2 PPH2 (S)-BINAP
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
a This compound cannot be completely planar because steric hindrance prevents the two ri...View the full answer

Answered By

Robert Mwendwa Nzinga
I am a professional accountant with diverse skills in different fields. I am a great academic writer and article writer. I also possess skills in website development and app development. I have over the years amassed skills in project writing, business planning, human resource administration and tutoring in all business related courses.
4.90+
187+ Reviews
378+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following two compounds. How would you distinguish between them using: a) IR spectroscopy? b) 1 H NMR spectroscopy? c) 13 C NMR spectroscopy?
-
Consider the following two compounds. Monochlorination of one of these compounds produces twice as many stereoisomeric products as the other. Draw the products in each case, and identify which...
-
Consider the following two compounds. When treated with NaOH, one of these compounds forms an epoxide quite rapidly, while the other forms an epoxide very slowly. Identify which compound reacts more...
-
A solid of constant density is bounded below by the plane z = 0, on the sides by the elliptical cylinder x 2 + 4y 2 = 4, and above by the plane z = 2 - x. a. Find x and y. b. Evaluate the integral...
-
Write an executive summary on "Fracking revolution puts America on course to export oil"
-
A magician has shaved an edge off one side of a six-sided die, and as a result, the die is no longer fair. The figure shows a graph of the probability density function (pdf). Show the pdf in table...
-
An element of a balanced orthotropic lamina is under the state of stress shown in Figure 4.11. The properties of the lamina are: Using the maximum strain criterion, determine whether or not failure...
-
Mega Games, Inc., produces two games, Extreme Combat and Judgment Weekend, with product structures as shown. An order for 100 units of Extreme Combat and 175 units of Judgment Weekend has been...
-
A tensile load is applied to a thin-walled hollow cylinder. Determine the change in wall thickness and in mean radius at the point of maximum load, if the stress curve is given by = Ae", where is the...
-
LEI has the following capital structure, which it considers to be optimal: Debt 25% Preferred stock 15 Common equity 60 100 LEIs expected net income this year is $34, 285.72, its established dividend...
-
Based on your analysis in the previous problem, determine whether the following allene is expected to be chiral: Me , Me, c=c=c Me
-
Using the data in the following table, predict the sign and magnitude of ÎH° for each of the following reactions. In each case, identify whether the reaction is expected to be endothermic...
-
What percent of students who took the test have a mathematics score above 680? Assume that the mathematics scores on a placement test are normally distributed with a mean of 500 and a standard...
-
Which of the following is most likely to cause the long-run aggregate supply curve to shift to the left? A. Higher nominal wages B. A decline in productivity C. An increase in corporate taxes
-
Deflation is most likely to be associated with: A. a shortage of government revenue. B. substantial macroeconomic contraction. C. explicit monetary policy to combat inflation.
-
What is the purpose of the Sarbanes-Oxley Act (SOX), and what are some of its provisions?
-
What is an accountable care organization (ACO), and what is it designed to accomplish?
-
The term that describes when inflation declines but nonetheless remains at a positive level is: A. deflation. B. stagflation. C. disinflation.
-
The Cobb-Douglas production function is given by f(x 1 , x 2 ) = Ax a 1 x b 2 . It turns out that the type of returns to scale of this function will depend on the magnitude of a+b. Which values of a...
-
Presented below are income statements prepared on a LIFO and FIFO basis for Kenseth Company, which started operations on January 1, 2024. The company presently uses the LIFO method of pricing its...
-
Show how you would prepare each of the following carboxylic acids through a Grignard synthesis: (a) (b) (c) (d) 4-Methylbenzoic acid (e) Hexanoic acid OH OH
-
(a) Which of the carboxylic acids in Practice Problem 17.6 could be prepared by a nitrile synthesis as well? In problem 17.6 (b) Which synthesis, Grignard or nitrile, would you choose to prepare OH ...
-
When maleic acid is heated to 200 8C, it loses water and becomes maleic anhydride. Practice Problem 17.8 Fumaric acid, a diastereomer of maleic acid, requires a much higher temperature before it...
-
Pietro Frozen Foods, Inc., produces frozen pizzas. For next year, Pietro predicts that 50,000 units will be produced, with the following total costs: Direct materials Direct labor Variable overhead...
-
You will complete two case studies over the course of the semester. These are to be completed individually. Each case study is found in the textbook and there are questions there to help guide your...
-
Thanasi has a business snowplowing. He uses a 2018 Ford Rapture with a snowplow outfitted to it. While plowing a driveway, he negligently ran it into the side of the neighbour's house. The damage to...

Study smarter with the SolutionInn App


