For each of the following compounds, determine whether the two protons shown in red are homotopic, enantiotopic,
Question:
(a)
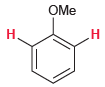
(b)
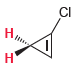
(c)
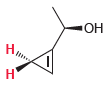
(d)
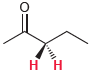
(e)
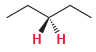
Discuss.
Transcribed Image Text:
OMe Н. Н CI H,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a Homotopic b ...View the full answer

Answered By

Ankur Gupta
I have a degree in finance from a well-renowned university and I have been working in the financial industry for over 10 years now. I have a lot of experience in financial management, and I have been teaching financial management courses at the university level for the past 5 years. I am extremely passionate about helping students learn and understand financial management, and I firmly believe that I have the necessary skills and knowledge to effectively tutor students in this subject.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following compounds, draw the important resonance forms. Indicate which structures are major and minor contributors or whether they have the same energy. (a) H2CNN (b) H2C=CH-NO2 (c)...
-
For each of the following compounds, give the systematic name and the common name (for those that have common names), and indicate whether the amines are primary, secondary, or tertiary: a. b. c....
-
For each of the following compounds, use the nitrogen rule to determine whether the molecular weight should be even or odd. Then calculate the expected m/z value for the molecular ion. a. b. c. d. O:...
-
Engineering is a dynamic field that requires continuous learning. Discuss how you plan to acquire and apply new knowledge as needed throughout your engineering career. Address the strategies you...
-
What is intellectual property? What are three examples of intellectual properties?
-
In the Spacelab Life Sciences 2, led by Paul X. Callahan, 14 male rats were sent to space. The red blood cell mass (in milliliters) of the rats was determined when they returned. A control group of...
-
Check whether the following can define probability distributions, and explain your answers. (a) \(f(x)=\frac{x}{10}\), for \(x=0,1,2,3,4\). (b) \(f(x)=\frac{1}{3}\), for \(x=-1,0,1\). (c)...
-
1. Take a poll of your class (at least, the Gen-X and Gen-Y members). At school or work, how many regularly (e.g., daily or every few days) send or receive information (not entertainment) using (a)...
-
Brite Toothbrushes has gathered the following Information to complete its Production Report for the month of April. Assume all materials are added at the beginning of the process. Required: Using the...
-
Suppose a distribution has Mean=20, Median=10, and SD=15. Then, the value of coefficient of skewness is, a) b) c) d) 1.50 2.00 -2.00 -3.00
-
Deduce the structure of a compound with molecular formula C 5 H 10 O that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. Data from the mass spectrum are also provided. 100 Mass Spec. Data...
-
Deduce the structure of a compound with molecular formula C 5 H 10 O that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. Data from the mass spectrum are also provided. 100 Mass Spec. Data...
-
Show that the internal rate of return of the following investment is 0, 100, and 200percent: Net investment Net cash flows -$1,000 +6,000 -11,000 +6.000 Year 0 Year l Year 2 Year 3
-
https://en.wikipedia.org/wiki/Concentration_(card_game) loops. Design a game with mutable world state, ArrayLists, mutable data structures and You will be using the Impworld library, as in Lab 8 -...
-
An individual's ethics is influenced by many factors. As a result, everyone's ethical reasoning differs. Companies and organizations create internal rules or codes of conducts to ensure consistent...
-
The bank reconciliation for Widgets Inc. included the following items: $30 bank service charge; NSF check of $275; bank error of $30; EFT collection from a customer for $2,335. Based on the...
-
York's outstanding stock consists of 80,000 shares of cumulative 7.5% preferred stock with a $5 par value and also 200,000 shares of common stock with a $1 par value. During its first four years of...
-
William acquires a $100,000 term life policy on his own life and then immediately assigns the policy to a registered charity. William plans on continuing to pay the $1,500 annual premium and is not...
-
Complete the requirements below using the most recent (20xx) financial statements available on Sonic Drive-Ins website. Obtain the statements on the Internet by following the steps below. (The...
-
1. Which of the four major types of information systems do you think is the most valuable to an organization? 2. How do you critically associate the ideas of business agility and business efficiency...
-
Normally, a Trans alkene is more stable than its cis isomer Trans-Cyclooctene, however, is less stable than cis-Cyclooctene by 38.5kJ/mol. Explain.
-
Trans-Cyclooctene is less stable than cis-Cyclooctene by 38.5kJ/mol, but Trans cyclononene is less stable than cis-cyclononene by only 12.2kJ/mol. Explain.
-
Allene (1, 2-propadiene), H2C = C = CH2, has two adjacent double bonds. What kind of hybridization must the central carbon have? Sketch the bonding orbitals in allene. What shape do you predict for...
-
1. Refer to the graph provided. Price, cost of unit $15- 9 MC ATC MR = P = D a. At what level of output does the firm maximize profit? Explain how you know. b. At the profit-maximizing quantity of...
-
A bond issued 10 years ago had a face value of $2,000; a coupon rate of 5%; and a yield of 6% when it was sold last month in the secondary bond market. At what price did the bond sell in the...
-
What are the assertions affected by the earlier list on what could go wrong in the post to the general journal process? The assertions to use are Completeness Existence/Occurrence Presentation and...

Study smarter with the SolutionInn App


