Draw the structure of the compound with molecular formula C 6 H 15 N that exhibits the
Question:
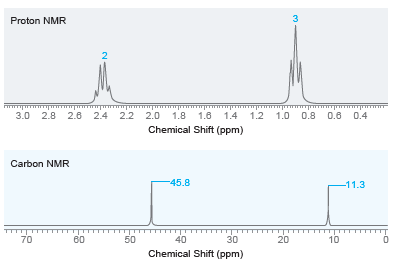
Transcribed Image Text:
3 Proton NMR 3.0 2.8 2.6 2.4 2.2 2.0 1.8 1.6 1.4 1.2 1.0 0.8 0.6 0.4 Chemical Shift (ppm) Carbon NMR -45.8 -11.3 40 10 50 70 80 30 20 Chemical Shift (ppm) 2.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)

Answered By

Allan Simiyu
I am an adroit Writer. I am a dedicated writer having worked as a writer for 3 years now. With this, I am sure to ace in the field by helping students break down abstract concepts into simpler ideas.
5.00+
8+ Reviews
54+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the structure of the compound with molecular formula C8H11N that exhibits the following 1 H NMR and 13 C NMR spectra: Proton NMR 2 22 Chemical Shift (ppm) Carbon NMR 128.8 128.4 40.0 -126.1...
-
Deduce the structure of a compound with molecular formula C 6 H 14 O 2 that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. 100 80 60 40 20 3000 2500 Wavenumber (cm-1) 4000 3500 2000 1500...
-
Deduce the structure of a compound with molecular formula C 6 H 14 O 2 that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. 100 80 60 40 20 4000 3500 3000 2500 2000 1500 1000 Wavenumber...
-
Perhaps more surprising to Mr. Pitkin was a proposal by the VP of Marketing to make a major investment in market share by increasing promotional expenditures by $2.5 million during 1998-2000. Sales...
-
Without calculating, determine whether the value of nPr is greater than the value of nCr for the values of n and r given in the table. Complete the table using yes (Y) or no (N). Is the value of nPr...
-
Do the payroll disbursement hours comply with Benfords Law?
-
Figure 2.3 displays the frequency distribution from Example 2.5. Create a similar plot using ggplot () to display the relative frequency distribution for each treatment group instead of the frequency...
-
The supply curve for product X is given by Qsx = - 520 + 20Px. a. Find the inverse supply curve. b. How much surplus do producers receive when Qx = 400? When Qx = 1,200?
-
relation R(A, B, C, D, E). given the following set F of functional dependencies for a F {ABC, BCD, EA, BEC, ABE D} = (a) Is BE ABC implied by F? In other words, is BE ABC F+? If yes, use the...
-
There is a lottery with n coupons and n people take part in it. Each person picks exactly one coupon. Coupons are numbered consecutively from 1 to n, n being the maximum ticket number. The winner of...
-
Using benzene as your only source of carbon atoms and ammonia as your only source of nitrogen atoms, propose a synthesis for the following compound: HN NH
-
Propose a mechanism for the following process: REN heat + N2 + CO2
-
Solve the linear programming problems in Problem using the simplex method. Maximize P = x1 + 2x2 + 3x3 subject to 2x1 + 2x2 + 8x3 < 600 X1 + 3x2 + 2x3 s 600 3x1 + 2x2 + X3 < 400 X1, X2, X3 2
-
You are working for a consumer rights organization. You are interested in knowing whether the milk contained in 16-oz (1-pint) bottles really weighs 16 oz. You do not want to accuse the packer of...
-
Identify the parent(s), subsidiaries and associates, if any, in the following structure: B 50% 60% A C D 60% 40%
-
Revenue is recognized in group accounts only when the control of inventory are transferred to a third party. True/False
-
Veronica manages a team of medical transcribers, who all work from home and are paid hourly. What potential problems might she have getting her remote team members to work as productively as she...
-
You are working at a marketing firm in ad sales. Your manager announces that the company is starting a new incentive program to increase the number of potential customers, or leads, for salespeople...
-
A hyperon decays as p + - ; its momentum in the L frame is p = 2 GeV. Take the direction of the in the L frame as the x-axis. In the CM frame the angle of the proton direction with x is * p =...
-
In a large midwestern university, 30% of the students live in apartments. If 200 students are randomly selected, find the probability that the number of them living in apartments will be between 55...
-
(a) Write the propagation steps leading to the formation of dichloromethane (CH2Cl2) from chloromethane. (b) Explain why free-radical halogenations usually gives mixtures of products. (c) How could...
-
Draw resonance forms to show how the BHA radical is stabilized by delocalization of the radical electron over other atoms in the molecule.
-
The triphenylmethyl cation is so stable that some of its salts can be stored for months. Explain why this cation is so stable. triphenylmethyl cation
-
Assume that Pope Enterprises held a $10,000, 10 percent, six-month note signed by Mary Drew. On December, 1, 2015, the maturity date, Drew dishonored the note. At this point, Drew owes a total of...
-
5. Consider the shape shown on the following graph and the brute force method of solving the convex hull problem 2 -2 -1 0 a. Label all the points that will be provided to the algorithm as input. You...
-
2. A project requires $150 million investment. If you invest today, the present value of future cash flow is worth $180 million. You can wait for one year to observe the demand of the market;...

Study smarter with the SolutionInn App


