Identify the weakest C!H bond in each of the following compounds: (a) (b) (c) (d)
Question:
Identify the weakest C!H bond in each of the following compounds:
(a)
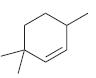
(b)
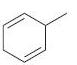
(c)
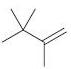
(d)
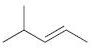
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
a...View the full answer

Answered By

Rustia Melrod
I am a retired teacher with 6 years of experience teaching various science subjects to high school students and undergraduate students. This background enables me to be able to help tutor students who are struggling with the science of business component of their education. Teaching difficult subjects has definitely taught me patience. There is no greater joy for me than to patiently guide a student to the correct answer. When a student has that "aha!" moment, all my efforts are worth it.
The Common Core standards are a useful yardstick for measuring how well students are doing. My students consistently met or exceeded the Common Core standards for science. I believe in working with each student's individual learning styles to help them understand the material. If students were struggling with a concept, I would figure out a different way to teach or apply that concept. I was voted Teacher of the Year six times in my career. I also won an award for Innovative Teaching Style at the 2011 National Teaching Conference.
4.90+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use the electro negatively table (Figure) to predict which bond in each of the following sets is more polar, and indicate the direction of bond polarity for each compound. (a) H3C ? C1 OR C1 ? C1 (b)...
-
Without using Fig. 13.3, predict which bond in each of the following groups is the most polar. a. COF, SiOF, GeOF b. POCl, SOCl c. SOF, SOCl, SOBr d. TiOCl, SiOCl, GeOCl e. COH, SiOH, SnOH f. AlOBr,...
-
Use the symbols δ+ and δ- to show the direction of polarity of the indicated bond in each of the following compounds (for example, a. HO--H b. F--Br c. H3C--NH2 d. H3C--Cl...
-
a. Over a two year time horizon an investor experiences the following net cash flows: Year 0 1 2 Cash flow -$8,000 $5,280 $5,320 What is the internal rate of return for the investor? Demonstrate that...
-
The contribution margin ratio of Thronson Corporation's only product is 61%. The company's monthly fixed expense is $456,300 and the company's monthly target profit is $42,300. Required: Determine...
-
Based on 107 students scores on the first examination in a course on business statistics, the following model was estimated by least squares: where y` = students actual score on the examination x1 =...
-
Water flows over a bump of height \(h=h(x)\) on the bottom of a wide rectangular channel as is indicated in Fig. P10.25. If energy losses are negligible, show that the slope of the water surface is...
-
Under the professional standards currently in effect, what responsibility do auditors have to identify internal control problems within their clients accounting systems? To whom must auditors...
-
What are some of the factors that determine health behaviors? Identify a health behavior that you struggle to perform? Which theory of health behavior change best relates to your inability to achieve...
-
Marys Luxury Travel uses a weekly federal income tax withholding table. Refer to Figure 8-4 in the text. The payroll data for each employee for the week ended March 22, 20, are given. Employees are...
-
Prove that C V = - (U/V) T (V/T) U.
-
Calculate the single-factor and multiple-factor productivities.
-
What are federal Crown corporation bonds? Which major Crown corporations issue these types of bonds? What characteristic makes these bonds especially attractive to investors?
-
Do you think concerns around data tracking in apps and approval of device access are justified? Search onlinecan you find any concerning downside examples of data breaches or harmful outcomes from...
-
Google is in an especially strong position when compared to other ad networks and online advertising competitors. Which sources of competitive advantage accrue to the firms that dominate ad networks?...
-
Search online to find estimates of the current highest paying cost per click. Which keywords pay the most? Why do you think firms are willing to spend so much?
-
Research the current state of Internet privacy law. Are laws consistent at the national and local levels? If not, what does this mean for firms operating in areas impacted by these laws?
-
Suppose the government amends the constitution to prevent government officials from negotiating with terrorists. What are the advantages of such a policy? What are the disadvantages?
-
Evaluate sin(a + 2x) 2 sin(a + x) + sin a lim x?
-
How is use of the word consistent helpful in fraud reports?
-
Classify each of the following monosaccharide?s: la) 0 (c) (b) (d) CH- . H- C=0 C=0 -- -- -- -- -- -- -- -- -- H2OH -- CH- Ribulose CH Threose CH- Tagatose 2-Deoxyribose
-
Convert the following Fischer projections into tetrahedral representations, and assign R or S stereochemistry toeach: H (b) (a) H2N- - (c) - - CH-CH CH CH
-
Which of the following Fischer projections of glyceraldehydes represent the sameenantiomer? CH- - 3 - -CH- - - CH2 A
-
3. Consider an economy in which the equilibrium solves the optimal growth problem 00 max B' (0 log(C)+(10) log(NhL,)) s.t.: t=0 C + K = (1 8)K, + AK (y'L)-a t C > 0 t+1 K>0 t+1 Ko is given 0 N = n'No...
-
Evaluate each limit. a) lim (x - 3x + 2) X-0 b) lim x2-x-2 X-2 x-2 c) lim x-3 X-9 X-9
-
If the variance of a probability was computed to be 3.6 grams, what is the standard deviation? Carlson Jewelers permits the return of their diamond wedding rings, provided the return occurs within...

Study smarter with the SolutionInn App


