Predict the products that are formed when diphenyl carbonate is treated with excess methyl magnesium bromide. 1)
Question:
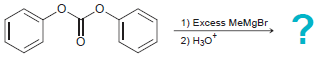
1) Excess MeMgBr 2) H3o*
Step by Step Answer:
1 exc...View the full answer



Related Video
The experiment aims to show the impact of various beverages on teeth by using eggs as a representation of enamel. Three eggs are boiled and then placed in glasses filled with fizzy drinks, vinegar, and mango juice for 24 hours. The shells of eggs are similar to enamel as they are composed of calcium carbonate, and enamel is primarily made of calcium phosphate. The eggs are then observed to demonstrate the effects of the different liquids on teeth and the importance of brushing regularly. The egg placed in fizzy drink has turned dark in color but can be cleaned by brushing with toothpaste and rinsing with water. The egg placed in vinegar has had its shell softened due to the chemical reaction of vinegar and calcium carbonate, which can\'t be reversed. This highlights the fact that acids are more damaging to teeth than other substances. The egg placed in mango juice represents the process of bacteria in the mouth converting sugars and starches into acids that form plaque, which can be prevented by brushing. The use of fluoride in toothpaste is also highlighted as it slows down the demineralization process and protects the enamel. The importance of brushing teeth twice a day is emphasized.
Students also viewed these Sciences questions
-
Predict the products that are expected when each of the following alkenes is treated with a peroxy acid (such as MCPBA) followed by aqueous acid: a. b. c. d. e. f.
-
Predict the products that are expected when each of the following alkenes is treated with ozone followed by DMS: a. b. c. d. e. f.
-
Predict the products that are expected when each of the following compounds is heated with concentrated HBr. a. b. c. d.
-
What are due process and stare decisis, and why are they important?
-
What are the strategically relevant factors of the macro-environment that affect the attractiveness of the U.S. jewelry industry? Specifically, are general and industry economic conditions and...
-
a. Is Jim acting professionally? Explain your response. b. Which of the four schools of ethical thought does Jim follow? Explain your response. c. Is the CFOs proposed action ethical?
-
The block is made of titanium Ti-6A1-4V and is subjected to a shortening of 0.06 in. along the y axis, and its shape is given a tilt of =89.7. Determine x , y , and Y xy . 4 in. 8 A 5 in.
-
On January 1, 2015, Geffrey Corporation had the following stockholders equity accounts. Common Stock ($20 par value, 60,000 shares issued and outstanding) ...$1,200,000 Paid-in Capital in Excess of...
-
An investment of $21700 is accumulated at 5.24% compounded quarterly for three and one-half years. At that time the interest rate is changed to 6.12% compounded monthly. How much is the investment...
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
What is poka -yoke, who developed the concept, and what do you think it has to do with lean?
-
What are the Five-Ss and from where did they originate?
-
Suppose X and Y are independent and Gaussian with means of X and Y, respectively, and equal variances of 2. The polar variables are formed according to R = X2 + Y2 and = tan1 (Y / X). - Find the...
-
What factors should a firm considering marketing its innovation in multiple countries use in formulating its protection strategy?
-
Write a Verilog description for the sequential circuit given by the state diagram in Figure 4-19(d). Include an asynchronous RESET signal to initialize the circuit to state Init. Compile your...
-
Pick a company you are familiar with. Can you identify some of its core competencies?
-
A sequential circuit has two lip- lops A and B, one input X, and one output Y. The state diagram is shown in Figure 4-54. Design the circuit with D lip- lops using a one- hot state assignment. Figure...
-
On the night of January 9, 2006, Mark Patterson, a police officer for the Westminster Police Department and the Internet Crimes Against Children Task Force, conducted an undercover investigation on...
-
Find a polynomial function (x) of least degree having only real coefficients and zeros as given. Assume multiplicity 1 unless otherwise stated. 0, -i, and 2 + i
-
Differentiate the following terms/concepts: a. Personality types and money attitudes b. Planners and avoiders c. Moderating and adapting to biases d. "Perfectible judges" and "incorrigible judges"
-
Use a Frost circle to determine the -electron structure of the cyclopropenyl cation, which has two electrons cyclopropenyl cation
-
Do you think it would be possible to have an aromatic free radical? Why or why not?
-
Which d the following species should be aromatic by the Huckel 4n + 2 rule? (a) (b) (c) N: isoxazole C2Hs
-
Given the function. f(x) = ln (2x+3x) Compute f'(x). Enter your answer rounded to the nearest tenth
-
A motor-cycle manufacturing company desires a profit of $600,000. The fixed costs are $800,000. The unit selling price of the motor-cycle is $750 and the variable cost per unit is $680. How many...
-
3. ZR Corporation's stock has a beta coefficient equal to 0.8 and a required rate of return equal to 11 percent. If the expected rate of return on the market is 12.5 percent, what is the risk-free...

Study smarter with the SolutionInn App


