3-Buten-2-one (3) has a strong absorption at 213nm and a weaker absorption at 320nm. Justify these features
Question:
3-Buten-2-one (3) has a strong absorption at 213nm and a weaker absorption at 320nm. Justify these features and assign the ultraviolet absorption transitions.
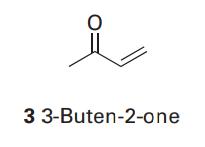
Transcribed Image Text:
3 3-Buten-2-one
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
Thorough explanation The 3buten2one molecule has two pos...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
The compound CH 3 CH=CHCHO has a strong absorption in the ultraviolet at 46 950 cm 1 and a weak absorption at 30 000 cm 1 . Justify these features in terms of the structure of the molecule.
-
Features of a strong internal control system Required List and describe nine features of a strong internal control system discussed in this chapter.
-
A compound A has a strong, broad IR absorption at 3200-3500 cm-1 and the proton NMR spectrum shown in Fig. P13.48a. Treatment of compound A with H2SO4 gives compound B, which has the NMR spectrum...
-
(a) An n n matrix K is the encryption matrix for the Hill cipher. Give the encryption formula. (b) The plaintext is (2, 5, 1,0). Encrypt it (show your work and the ciphertext) using the Hill cipher...
-
What is meant by the term normal balance? Provide the normal balance for assets, liabilities, and stockholders equity.
-
The Stahl Metalworks Corp. general ledger contained the following information at September 30, 2020: Cost of goods...
-
Johanna Marra and Eric Nazzaro began a romantic relationship in October 2013. That previous July, Nazzarro had purchased a duplex that he intended to renovate. Nazzarro rented out the top floor while...
-
Hart Computer Company has been purchasing carrying cases for its portable computers at a delivered cost of $68 per unit. The company, which is currently operating below full capacity, charges factory...
-
Jobs R Us, Inc. Is a recruiting firm that specializes in post- college placement in the finance industry. Its clients are currently concentrated in the North-Eastern United States lt is contemplating...
-
Lakeside Bakery bakes fresh pies every morning. The daily demand for its apple pies is a random variable with (discrete) distribution, based on past experience, given by Each apple pie costs the...
-
Suppose that we can model a charge-transfer transition in a onedimensional system as a process in which a Gaussian wavefunction centred on x=0 and width a makes a transition to another Gaussian...
-
Figure 13.2 shows the UV-visible absorption spectra of a selection of amino acids. Suggest reasons for their different appearances in terms of the structures of the molecules. Data in Figure 13.2...
-
If Saturn could fit in a bathtub, would it float? Use the planetary data in Appendix A to calculate the density (mass/volume) of Earth, Mars, Jupiter, and Saturn. What are the differences? Can you...
-
Using the completing-the-square method, find the vertex of the function f(x)=-2x^(2)+12x+5 and indicate whether it is a minimum or a maximum.
-
Proton Limited acquired the net assets and contingent liabilities of Electron Limited for $70 000. Electron Limited's net assets and contingent liabilities were: total assets $94 000; total...
-
1. Identify 2 types of financial loss from a crisis event. ( Note that a financial loss is not the same as a crisis in a financial market.) Provide an example of each type of loss. Which one of your...
-
A 0.50 kg object is attached to a spring with force constant 169 N/m so that the object is allowed to move on a horizontal frictionless surface. The object is released from rest when the spring is...
-
What is the value of egg_count after running this code? egg_count = 0 def buy_eggs(): egg_count += 12 buy_eggs()
-
What are the advantages and disadvantages of computer interactive interviewing compared to the traditional methods of surveying?
-
The electric field due to a line charge is given by where l is a constant. Show that E is solenoidal. Show that it is also conservative. E =
-
Why didnt Joule change his experiment to make C surroundings /C system approx = 0.001 to increase the sensitivity of the apparatus?
-
Why does the relation C P > C V always hold for a gas? Can C P < C V be valid for a liquid?
-
Why can q V be equated with a state function if q is not a state function?
-
Choose an environmental law or policy (please don't choose Clean Air Act...challenge yourselves). Why did the legislation/policy come about, and what has been its effect on the environment ?
-
Although Malaysia has made a concerted effort to preserve and enhance environmental quality, it is still confronted with the competing demands of greater urbanisation and environmental preservation. ...
-
Respond to the following questions: 1. Characterize the elements of vehicle insurance coverage by addressing the following: a. Describe the type of coverage provided under each of the four "Parts" of...

Study smarter with the SolutionInn App


